[three_fourth last=”no”]
[tabs tab1=”2020-Present” tab2=”2010-2019″ tab3=”2006-2009″ layout=”horizontal” backgroundcolor=”” inactivecolor=””]
[tab id=1]
[accordian]
[toggle title=”Fluorescent Azobenzene-confined Coiled-coil Mesofibers” open=”yes”]
[title size=”3″]Abstract[/title]
Fluorescent protein biomaterials have important applications such as bioimaging in pharmacological studies. Self-assembly of proteins, especially into fibrils, is known to produce fluorescence in the blue band. Capable of self-assembly into nanofibers, we have shown we can modulate its aggregation into mesofibers by encapsulation of a small hydrophobic molecule. Conversely, azobenzenes are hydrophobic small molecules that are virtually non-fluorescent in solution due to their highly efficient photoisomerization. However, they demonstrate fluorogenic properties upon confinement in nanoscale assemblies by reducing the non-radiative photoisomerization. Here, we report the fluorescence of a hybrid protein-small molecule system in which azobenzene is confined in our protein assembly leading to fiber thickening and increased fluorescence. We show our engineered protein Q encapsulates AzoCholine, bearing a photoswitchable azobenzene moiety, in the hydrophobic pore to produce fluorescent mesofibers. This study further investigates the photocontrol of protein conformation as well as fluorescence of an azobenze-containing biomaterial.
[title size=”4″]Citation[/title]
Punia, K.; Britton, D.; Hüll, K.; Yin, L.; Wang, Y.; Renfrew, P. D.; Gilchrist, M. L.; Bonneau, R.; Trauner, D.; Montclare, J. K. Fluorescent Azobenzene-confined Coiled-coil Mesofibers. Soft Matter 2023, XX, XX-XX. doi: 10.1039/D2SM01578A
[/toggle]
[toggle title=”Protein-Engineered Biomaterials for Cartilage Therapeutics and Repair” open=”no”]
[title size=”3″]Abstract[/title]
Cartilage degeneration and injury are major causes of pain and disability that effect millions, and yet treatment options for conditions like osteoarthritis (OA) continue to be mainly palliative or involve complete replacement of injured joints. Several biomaterial strategies have been explored to address cartilage repair either by the delivery of therapeutics or as support for tissue repair, however the complex structure of cartilage tissue, its mechanical needs, and lack of regenerative capacity have hindered this goal. Recent advances in synthetic biology have opened new possibilities for engineered proteins to address these unique needs. Engineered protein and peptide-based materials benefit from inherent biocompatibility and nearly unlimited tunability as they utilize the body’s natural building blocks to fabricate a variety of supramolecular structures. The pathophysiology and needs of OA cartilage are presented here, along with an overview of the current state of the art and next steps for protein-engineered repair strategies for cartilage.
[title size=”4″]Citation[/title]
Haq-Siddiqi, N. A.; Britton, D.; Montclare, J. K. Protein-Engineered Biomaterials for Cartilage Therapeutics and Repair. Adv. Drug Deliv. Rev., 2022, XX, XX-XX. doi: 10.1016/j.addr.2022.114647
[/toggle]
[toggle title=”Tuning a Coiled-coil Hydrogel via Computational Design of Supramolecular Fiber Assembly” open=”no”]
[title size=”3″]Abstract[/title]
Previously reported, Q, is a thermoresponsive coiled-coil protein capable of higher-order supramolecular assembly into fibers and hydrogels with upper critical solution temperature (UCST) behavior. Here, we introduce a new coiled-coil protein, Q2, that is redesigned to favor longitudinal growth over lateral growth of its fibers and thus achieve a higher crosslinking density within the formed hydrogel. We also introduce a favorable hydrophobic mutation to the pore of the coiled-coil domain for increased thermostability of the protein. We note that an increase in storage modulus of the hydrogel and crosslinking density is coupled with a decrease in fiber diameter. We further fully characterize our α-helical coiled-coil Q2 hydrogel for its structure, nano-assembly, and rheology relative to our previous single domain protein, Q, over the time of its gelation demonstrating the nature of our hydrogel self-assembly system. The design parameters here not only show the importance of electrostatic potential in self-assembly but provide a step towards predictable design of electrostatic protein interactions.
[title size=”4″]Citation[/title]
Britton, D.; Meleties, M.; Liu, C.; Jia, S.; Mahmoudinobar, F.; Renfrew, P. D.; Bonneau, R.; Montclare, J. K. Tuning a Coiled-coil Hydrogel via Computational Design of Supramolecular Fiber Assembly. Mol. Syst. Des. Eng., 2022, XX, XX-XX. doi: 10.1039/D2ME00153E
[/toggle]
[toggle title=”Supramolecular Assembly and Small-Molecule Binding by Protein-Engineered Coiled-Coil Fibers” open=”no”]
[title size=”3″]Abstract[/title]
The ability to engineer a solvent-exposed surface of self-assembling coiled coils allows one to achieve a higher-order hierarchical assembly such as nano- or microfibers. Currently, these materials are being developed for a range of biomedical applications, including drug delivery systems; however, ways to mechanistically optimize the coiled-coil structure for drug binding are yet to be explored. Our laboratory has previously leveraged the functional properties of the naturally occurring cartilage oligomeric matrix protein coiled coil (C), not only for its favorable motif but also for the presence of a hydrophobic pore to allow for small-molecule binding. This includes the development of Q, a rationally designed pentameric coiled coil derived from C. Here, we present a small library of protein microfibers derived from the parent sequences of C and Q bearing various electrostatic potentials with the aim to investigate the influence of higher-order assembly and encapsulation of candidate small molecule, curcumin. The supramolecular fiber size appears to be well-controlled by sequence-imbued electrostatic surface potential, and protein stability upon curcumin binding is well correlated to relative structure loss, which can be predicted by in silico docking.
[title size=”4″]Citation[/title]
Britton, D.; Monkovic, J.; Jia, S.; Liu, C.; Mahmoudinobar, F.; Meleties, M.; Renfrew, P. D.; Bonneau, R.; Montclare, J. K. Supramolecular Assembly and Small-Molecule Binding by Protein-Engineered Coiled-Coil Fibers. Biomacromolecules 2022, 23 (11), 4851–4859. doi: 10.1021/acs.biomac.2c01031
[/toggle]
[toggle title=”Gendered Perception of Online University Learning of STEM Entrepreneurship during the COVID-19 Pandemic” open=”no”]
[title size=”3″]Abstract[/title]
Online educational experiences have exploded in popularity at the advent of the internet of things. Just as all forms before it, online learning has experienced implicit biases. With the abrupt lockdowns mandated by governments across the world, as a result of the COVID-19 pandemic, many educational institutions were thrust into an online learning environment with similarities and differences to the established online institutions already in place. Educational systems were forced to prepare and install methods of communication and instruction by using online resources such as online video calls and discussion forums. Here, we assess the impact of the COVID-19 pandemic on the Pre-Capstone Innovation Experience course at New York University, which provides entrepreneurship education in science, technology, engineering, and math (STEM) for undergraduates. Assessments captured between 2017 and 2020 from Pre-Capstone Innovation Experience participants demonstrate the gendered impact of the COVID-19 pandemic on the learning of undergraduate students.
[title size=”4″]Citation[/title]
Britton, D.; Thermer, S.; Perez, J.; Montclare, J. K. Gendered Perception of Online University Learning of STEM Entrepreneurship during the COVID-19 Pandemic. Technology & Innovation 2022, 22, 1-9. doi: 10.21300/22.3.2022.3 [/toggle]
[toggle title=”Fluorinated Protein and Peptide Materials for Biomedical Applications” open=”no”]
[title size=”3″]Abstract[/title]
Fluorination represents one of the most powerful modern design strategies to impart biomacromolecules with unique functionality, empowering them for widespread application in the biomedical realm. However, the properties of fluorinated protein materials remain unpredictable due to the heavy context-dependency of the surrounding atoms influenced by fluorine’s strong electron-withdrawing tendencies. This review aims to discern patterns and elucidate design principles governing the biochemical synthesis and rational installation of fluorine into protein and peptide sequences for diverse biomedical applications. Several case studies are presented to deconvolute the overgeneralized fluorous stabilization effect and critically examine the duplicitous nature of the resultant enhanced chemical and thermostability as it applies to use as biomimetic therapeutics, drug delivery vehicles, and bioimaging modalities.
[title size=”4″]Citation[/title]
Monkovic, J. M.; Gibson, H.; Sun, J. W.; Montclare, J. K. Fluorinated Protein and Peptide Materials for Biomedical Applications. Pharmaceuticals 2022, 15 (10), 1201. doi: 10.3390/ph15101201 [/toggle]
[toggle title=”Engineered multivalent self-assembled binder protein against SARS-CoV-2 RBD” open=”no”]
[title size=”3″]Abstract[/title]
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has caused a global pandemic since December 2019, and with it, a push for innovations in rapid testing and neutralizing antibody treatments in an effort to solve the spread and fatality of the disease. One such solution to both of these prevailing issues is targeting the interaction of SARS-CoV-2 spike receptor binding domain (RBD) with the human angiotensin-converting enzyme 2 (ACE2) receptor protein. Structural studies have shown that the N-terminal alpha-helix comprised of the first 23 residues of ACE2 plays an important role in this interaction. Where it is typical to design a binding domain to fit a target, we have engineered a protein that relies on multivalency rather than the sensitivity of a monomeric ligand to provide avidity to its target by fusing the N-terminal helix of ACE2 to the coiled-coil domain of the cartilage oligomeric matrix protein. The resulting ACE-MAP is able to bind to the SARS-CoV-2 RBD with improved binding affinity, is expressible in E. coli, and is thermally stable and relatively small (62 kDa). These properties suggest ACE-MAP and the MAP scaffold to be a promising route towards developing future diagnostics and therapeutics to SARS-CoV-2.
[title size=”4″]Citation[/title]
Britton, D.; Punia, K.; Mahmoudinobar, F.; Tada, T.; Jiang, X.; Renfrew, P. D.; Bonneau, R.; Landau, N. R.; Kong, X. -P.; Montclare, J. K. Engineered multivalent self-assembled binder protein against SARS-CoV-2 RBD. Biochem. Eng. J. 2022, 187, 108596. doi: 10.1016/j.bej.2022.108596 [/toggle]
[toggle title=”Bioinspired Green Science and Technology Symposium in NYC” open=”no”]
[title size=”3″]Abstract[/title]
In observance of Earth Day 2022 and the looming, urgent need to fight climate change and biodiversity loss, scientists gathered in New York City and online for the Bioinspired Green Science and Technology Symposium to share the latest technological and design breakthroughs that hold promise for the environment and human health.
[title size=”4″]Citation[/title]
Alcantar, N. A.; Banta, S.; Cak, A. D.; Chen, X.; DelRe, C.; Deravi, L. F.; Dordick, J. S.; Giebel, B. M.; Greenfield, D.; Groffman, P. M.; Holford, M.; John, G.; Joshi, N. S.; Kotov, N. A.; Montclare, J. K.; Moore, B. S.; Ortony, J. H.; Reinmann, A. B.; Wilson, C. J. Bioinspired Green Science and Technology Symposium in NYC. Matter 2022, 5 (7), 1980-1984. doi: 10.1016/j.matt.2022.06.031 [/toggle]
[toggle title=”Engineered protein–iron oxide hybrid biomaterial for MRI-traceable drug encapsulation” open=”no”]
[title size=”3″]Abstract[/title]
Labeled protein-based biomaterials have become popular for various biomedical applications such as tissue-engineered, therapeutic, and diagnostic scaffolds. Labeling of protein biomaterials, including with ultrasmall superparamagnetic iron oxide (USPIO) nanoparticles, has enabled a wide variety of imaging and therapeutic techniques. These USPIO-based biomaterials are widely studied in magnetic resonance imaging (MRI), thermotherapy, and magnetically-driven drug delivery, which provide a method for direct and non-invasive monitoring of implants or drug delivery agents. Where most developments have been made using polymers or collagen hydrogels, shown here is the use of a rationally designed protein as the building block for a meso-scale fiber. While USPIOs have been chemically conjugated to antibodies, glycoproteins, and tissue-engineered scaffolds for targeting or improved biocompatibility and stability, these constructs have predominantly served as diagnostic agents and often involve harsh conditions for USPIO synthesis. Here, we present an engineered protein–iron oxide hybrid material comprised of an azide-functionalized coiled-coil protein with small molecule binding capacity conjugated via bioorthogonal azide–alkyne cycloaddition to an alkyne-bearing iron oxide templating peptide, CMms6, for USPIO biomineralization under mild conditions. The coiled-coil protein, dubbed Q, has been previously shown to form nanofibers and, upon small molecule binding, further assembles into mesofibers via encapsulation and aggregation. The resulting hybrid material is capable of doxorubicin encapsulation as well as sensitive T₂*-weighted MRI darkening for strong imaging capability that is uniquely derived from a coiled-coil protein.
[title size=”4″]Citation[/title]
Hill, L. K.; Britton, D.; Jihad, T.; Punia, K.; Xie, X.; Delgado-Fukushima, E.; Liu, C. F.; Mishkit, O.; Liu, C.; Hu, C.; Meleties, M.; Renfrew, P. D.; Bonneau, R.; Wadghiri, Y. Z.; Montclare, J. K. Engineered protein–iron oxide hybrid biomaterial for MRI-traceable drug encapsulation. Mol. Syst. Des. Eng., 2022, 7, 915-932. doi: 10.1039/D2ME00002D [/toggle]
[toggle title=”Exploring the Viability and Role of Virtual Laboratories in Chemistry Education Using Two Original Modules” open=”no”]
[title size=”3″]Abstract[/title]
Virtual chemistry laboratory modules were investigated as a potential learning resource that complements traditional in-person experiments. In collaboration with a high school instructor, we designed two interactive modules, Rates of Reaction and Models of Organic Molecules, which became part of a previously reported online chemistry platform referred to as LabLessons. High school students took advantage of these modules to reinforce their knowledge of specific chemistry concepts and prepare themselves for the corresponding lab experiments. Students and the instructor were surveyed after their use of the two modules, providing feedback on the module designs and effectiveness. Survey results as well as academic performance suggest that independent learning through modules had an overall positive impact on learning when used in conjunction with traditional methods.[title size=”4″]Citation[/title]
Qu, A.; Nicolas M.; Leung, E. M.; Jones, S. M.; Katyal, P.; Punia, K.; Maxfield, M.; Montclare, J. K. Exploring the Viability and Role of Virtual Laboratories in Chemistry Education Using Two Original Modules. J. Chem. Educ. 2022, 99, 4, 1596-1603. doi: 10.1021/acs.jchemed.1c00892 [/toggle]
[toggle title=”Particle-Based Microrheology As a Tool for Characterizing Protein-Based Materials” open=”no”]
[title size=”3″]Abstract[/title]
Microrheology based on video microscopy of embedded tracer particles has the potential to be used for high-throughput protein-based materials characterization. This potential is due to a number of characteristics of the techniques, including the suitability for measurement of low sample volumes, noninvasive and noncontact measurements, and the ability to set up a large number of samples for facile, sequential measurement. In addition to characterization of the bulk rheological properties of proteins in solution, for example, viscosity, microrheology can provide insight into the dynamics and self-assembly of protein-based materials as well as heterogeneities in the microenvironment being probed. Specifically, passive microrheology in the form of multiple particle tracking and differential dynamic microscopy holds promise for applications in high-throughput characterization because of the lack of user interaction required while making measurements. Herein, recent developments in the use of multiple particle tracking and differential dynamic microscopy are reviewed for protein characterization and their potential to be applied in a high-throughput, automatable setting.
[title size=”4″]Citation[/title]
Meleties, M.; Martineau, R. L.; Gupta, M. K.; Montclare, J. K. Particle-Based Microrheology as a Tool for Characterizing Protein-Based Materials, ACS Biomater. Sci. and Eng. 2022, 8 (7), 2747–2763. doi: 10.1021/acsbiomaterials.2c00035 [/toggle]
[toggle title=”Smart Nanofibrous Materials for Tissue Engineering. In Biomimetic Protein Based Elastomers: Emerging Materials for the Future ” open=”no”]
[title size=”3″]Abstract[/title]
Tissue engineering is an attractive field of biomedicine directed toward creating temporary functional scaffolds in lieu of impaired tissues and organs. A fundamental challenge in tissue engineering is to mimic the molecular architecture and complexity of the extracellular matrix. Nanofibrous scaffolds have garnered significant attention in tissue engineering due to their high surface-area-to-volume ratio and porosity crucial for cell attachment, migration, and proliferation. A ‘smart’ material bears key attributes, including responsiveness to stimuli and the ability to self-assemble. Recent advances in smart nanofibrous materials pave the way to generate biomimetic scaffolds with structural integrity and a conducive environment for the cells to proliferate into a functional tissue. Here we review biomedical advances in protein and synthetic polymer-based smart nanofibers for tissue engineering applications.
[title size=”4″]Citation[/title]
Punia, K.; Bhattacharya, A.; Montclare, J. K. Smart Nanofibrous Materials for Tissue Engineering. In Biomimetic Protein Based Elastomers: Emerging Materials for the Future; Royal Society of Chemistry: London, UK, 2022; pp 173-209. doi: 10.1039/9781788012720-00173 [/toggle]
[toggle title=”High-Throughput Microrheology for the Assessment of Protein Gelation Kinetics” open=”no”]
[title size=”3″]Abstract[/title]
A high-throughput microrheological assay is employed to assess the gelation kinetics of a coiled-coil protein, Q, across a compositional space with varying ionic strengths and pH values. Two methods of passive microrheology─multiple particle tracking (MPT) and differential dynamic microscopy (DDM)─are used to determine mean-squared displacements of tracer beads embedded in protein solutions with respect to lag time over a fixed period. MPT data was analyzed to determine gelation kinetics in a high-throughput, automatable manner by fitting relaxation exponents to sigmoidal curves and verifying with the more traditionally used time-cure superposition. DDM-determined gelation time was assessed as the last resolvable time, which we found to be on a similar scale to gelation times given by MPT. Both methods show distinct advantages with regard to being used in a high-throughput, automatable setup; DDM can serve as an effective initial screen for rapid gelation kinetics due to it requiring less user intervention and inputs, with MPT giving a more complete understanding of the entire gelation process. Using these methods, a clear optimum for rapid gelation was observed near the isoelectric point of Q and at higher ionic strengths over the compositional space studied.
[title size=”4″]Citation[/title]
Meleties, M.; Britton, D.; Katyal, P.; Lin, B.; Martineau, R. L.; Gupta, M. K.; Montclare, J. K. High-Throughput Microrheology for the Assessment of Protein Gelation Kinetics. Macromolecules 2022, 55 (4), 1239–1247. doi: 10.1021/acs.macromol.1c02281 [/toggle]
[toggle title=”Injectable recombinant block polymer gel for sustained delivery of therapeutic protein in post traumatic osteoarthritis” open=”no”]
[title size=”3″]Abstract[/title]
Background:Protein-based biomaterials offer several advantages over synthetic materials, owing to their unique stimuli-responsive properties, biocompatibility and modular nature. Here, we demonstrate that E5C, a recombinant protein block polymer, consisting of five repeats of elastin like polypeptide (E) and a coiled-coil domain of cartilage oligomeric matrix protein (C), is capable of forming a porous networked gel at physiological temperature, making it an excellent candidate for injectable biomaterials. Combination of E5C with Atsttrin, a chondroprotective engineered derivative of anti-inflammatory growth factor progranulin, provides a unique biochemical and biomechanical environment to protect against post-traumatic osteoarthritis (PTOA) onset and progression. E5C gel was demonstrated to provide prolonged release of Atsttrin and inhibit chondrocyte catabolism while facilitating anabolic signaling in vitro. We also provide in vivo evidence that prophylactic and therapeutic application of Atsttrin-loaded E5C gels protected against PTOA onset and progression in a rabbit anterior cruciate ligament transection model. Collectively, we have developed a unique protein-based gel capable of minimally invasive, sustained delivery of prospective therapeutics, particularly the progranulin-derivative Atsttrin, for therapeutic application in OA.
[title size=”4″]Citation[/title]
Katyal, P.; Hettinghouse, A.; Meleties, M.; Hasan, S.; Chen, C.; Cui, M.; Sun, G.; Menon, R.; Lin, B.; Regatte, R.; Montclare, J. K.; Liu, C. Injectable recombinant block polymer gel for sustained delivery of therapeutic protein in post traumatic osteoarthritis. Biomaterials 2022, 281, 121370. doi: 10.1016/j.biomaterials.2022.121370 [/toggle]
[toggle title=”Protein-based lateral flow assays for COVID-19 detections” open=”no”]
[title size=”3″]Abstract[/title]
To combat the enduring and dangerous spread of COVID-19, many innovations to rapid diagnostics have been developed based on proteinprotein interactions of the SARS-CoV-2 spike and nucleocapsid proteins to increase testing accessibility. These antigen tests have most prominently been developed using the lateral flow assay (LFA) test platform which has the benefit of administration at point-of-care, delivering quick results, lower cost, and does not require skilled personnel. However, they have gained criticism for an inferior sensitivity. In the last year, much attention has been given to creating a rapid LFA test for detection of COVID-19 antigens that can address its high limit of detection while retaining the advantages of rapid antibodyantigen interaction. In this review, a summary of these proteinprotein interactions as well as the challenges, benefits, and recent improvements to protein based LFA for detection of COVID-19 are discussed.
[title size=”4″]Citation[/title]
Mahmoudinobar, F.; Britton, D.; Montclare, J. K. Protein-based lateral flow assays for COVID-19 detection, Protein Eng. Des. Sel. 2021, 34, gzab010. doi:10.1093/protein/gzab010 [/toggle]
[toggle title=”Protein based biomaterials for therapeutic and diagnostic applications” open=”No”]
[title size=”3″]Abstract[/title]
Proteins are some of the most versatile and studied macromolecules with extensive biomedical applications. The natural and biological origin of proteins offer such materials several advantages over their synthetic counterparts, such as innate bioactivity, recognition by cells and reduced immunogenic potential. Furthermore, proteins can be easily functionalized by altering their primary amino acid sequence and can often be further self-assembled into higher order structures either spontaneously or under specific environmental conditions. This review will feature the recent advances in protein-based biomaterials in the delivery of therapeutic cargo such as small molecules, genetic material, proteins, and cells. First, we will discuss the ways in which secondary structural motifs, the building blocks of more complex proteins, have unique properties that enable them to be useful for therapeutic delivery. Next, supramolecular assemblies, such as fibers, nanoparticles, and hydrogels, made from these building blocks that are engineered to behave in a cohesive manner, are discussed. Finally, we will cover additional modifications to protein materials that impart environmental responsiveness to materials. This includes the emerging field of protein molecular robots, and relatedly, protein-based theranostic materials that combine therapeutic potential with modern imaging modalities, including near-infrared fluorescence spectroscopy (NIRF), single-photo emission computed tomography/computed tomography (SPECT/CT), positron emission tomography (PET), magnetic resonance imaging (MRI), and ultrasound/photoacoustic imaging (US/PAI).
[title size=”4″]Citation[/title]
Chu, S.; Wang, A. L.; Bhattacharya, A.; Montclare, J. K. Protein based biomaterials for therapeutic and diagnostic applications. Prog. Biomed. Eng. 2022, 4, 012003.
doi: 10.1088/2516-1091/ac2841 [/toggle]
[toggle title=”Pollen-inspired enzymatic microparticles to reduce organophosphate toxicity in managed pollinators” open=”no”]
[title size=”3″]Abstract[/title]
Pollinators support the production of the leading food crops worldwide. Organophosphates are a heavily used group of insecticides that pollinators can be exposed to, especially during crop pollination. Exposure to lethal or sublethal doses can impair fitness of wild and managed bees, risking pollination quality and food security. Here we report a low-cost, scalable in vivo detoxification strategy for organophosphate insecticides involving encapsulation of phosphotriesterase (OPT) in pollen-inspired microparticles (PIMs). We developed uniform and consumable PIMs capable of loading OPT at 90% efficiency and protecting OPT from degradation in the pH of a bee gut. Microcolonies of Bombus impatiens fed malathion-contaminated pollen patties demonstrated 100% survival when fed OPT−PIMs but 0% survival with OPT alone, or with plain sucrose within five and four days, respectively. Thus, the detrimental effects of malathion were eliminated when bees consumed OPT−PIMs. This design presents a versatile treatment that can be integrated into supplemental feeds such as pollen patties or dietary syrup for managed pollinators to reduce risk of organophosphate insecticides.
[title size=”4″]Citation[/title]
Chen, J.; Webb, J.; Shariati, K.; Guo, S.; Montclare, J. K.; McArt, S.; Ma, M. Pollen-inspired enzymatic microparticles to reduce organophosphate toxicity in managed pollinators. Nat. Food 2021, 2, 339–347. doi: 10.1038/s43016-021-00282-0 [/toggle]
[toggle title=”Self-assembly of stimuli-responsive coiled-coil fibrous hydrogels” open=”no”]
[title size=”3″]Abstract[/title]
Towing to their tunable properties, hydrogels comprised of stimuli-sensitive polymers are one of the most appealing scaffolds with applications in tissue engineering, drug delivery and other biomedical fields. We previously reported a thermoresponsive hydrogel formed using a coiled-coil protein, Q. Here, we expand our studies to identify the gelation of Q protein at distinct pH conditions, creating a protein hydrogel system that is sensitive to temperature and pH. Through secondary structure analysis, transmission electron microscopy, and rheology, we observed that Q self-assembles and forms fiber-based hydrogels exhibiting upper critical solution temperature behavior with increased elastic properties at pH 7.4 and pH 10. At pH 6, however, Q forms polydisperse nanoparticles, which do not further self-assemble and undergo gelation. The high net positive charge of Q at pH 6 creates significant electrostatic repulsion, preventing its gelation. This study will potentially guide the development of novel scaffolds and functional biomaterials that are sensitive towards biologically relevant stimuli.
[title size=”4″]Citation[/title]
Meleties, M.; Katyal, P.; Lin, B.; Britton, D.; Montclare, J. K. Self-assembly of stimuli-responsive coiled-coil fibrous hydrogels. Soft Matter 2021, 17, 6470–6476. doi: 10.1039/D1SM00780G [/toggle]
[toggle title=”Enzyme Catalyzed Hydrolysis of Synthetic Polymers” open=”no”]
[title size=”3″]Abstract[/title]
Traditional plastics pose a grave threat to the environment with several hundred million tons of plastics being accumulated as waste. With conventional approaches requiring extreme temperatures and harsh conditions, degradation or recyclability achieved via enzymatic methods offer a meaningful alternative. In this chapter, we discuss enzymes that have been utilized for polymer degradation. We highlighted strategies to improve stability and catalytic efficiencies of these enzymes via protein engineering or anchoring peptides/domains to the catalytic domain or using additives that can enhance the binding of these enzymes to polymeric surfaces.
[title size=”4″]Citation[/title]
Katyal, P. and Montclare, J. K. Chapter 3: Enzyme Catalyzed Hydrolysis of Synthetic Polymers. In Sustainability & Green Polymer Chemistry Volume 2: Biocatalysis and Biobased Polymers; American Chemical Society: Washington D.C., USA, 2020; pp 47–63. doi: 10.1021/bk-2020-1373.ch003 [/toggle]
[toggle title=”Effect of Divalent Metal Cations on the Conformation, Elastic Behavior, and Controlled Release of a Photocrosslinked Protein Engineered Hydrogel” open=”no”]
[title size=”3″]Abstract[/title]
The fabrication of protein hydrogels consisting of different properties and functional motifs is critical in the development of protein-based materials for biomedical applications. Here, we report the design and characterization of a triblock protein polymer, CEC, composed of two different self-assembling domains derived from elastin protein (E) and coiled-coil protein (C), photopolymerized with a NHS-diazirine (D) crosslinker into a CEC-D hydrogel. The optimal photocrosslinker concentration and exposure time is determined to fabricate a free-standing hydrogel. Upon increasing the concentration of the CEC-D monomer and environmental temperature, the CEC-D hydrogel’s conformation decreases in helical content from 58.0% to 44.8% and increases in β-content from 25.9% to 38.1%. These gels experience 55 ± 6% protein erosion from the free-standing gel in 13 days as the gel films gradually decrease in size. The swelling ratio of 12 ± 1% denotes that the gel has a swelling ability comparable to other protein hydrogels. These photocrosslinked CEC-D hydrogels can be employed for drug delivery with high encapsulation and 14 ± 2% release of curcumin into the supernatant in a week long study. Overall, the photocrosslinked CEC-D hydrogels exhibit stability, swelling ability, and sustained release of drugs.
[title size=”4″]Citation[/title]
Wang, Y.; Wang, X.; Montclare, J. K. Effect of Divalent Metal Cations on the Conformation, Elastic Behavior, and Controlled Release of a Photocrosslinked Protein Engineered Hydrogel. ACS Appl. Bio Mater. 2021, 4 (4), 3587–3597. doi: 10.1021/acsabm.1c00091 [/toggle]
[toggle title=”Free-Standing Photocrosslinked Protein Polymer Hydrogels for Sustained Drug Release” open=”no”]
[title size=”3″]Abstract[/title]
The fabrication of protein hydrogels consisting of different properties and functional motifs is critical in the development of protein-based materials for biomedical applications. Here, we report the design and characterization of a triblock protein polymer, CEC, composed of two different self-assembling domains derived from elastin protein (E) and coiled-coil protein (C), photopolymerized with a NHS-diazirine (D) crosslinker into a CEC-D hydrogel. The optimal photocrosslinker concentration and exposure time is determined to fabricate a free-standing hydrogel. Upon increasing the concentration of the CEC-D monomer and environmental temperature, the CEC-D hydrogel’s conformation decreases in helical content from 58.0% to 44.8% and increases in β-content from 25.9% to 38.1%. These gels experience 55 ± 6% protein erosion from the free-standing gel in 13 days as the gel films gradually decrease in size. The swelling ratio of 12 ± 1% denotes that the gel has a swelling ability comparable to other protein hydrogels. These photocrosslinked CEC-D hydrogels can be employed for drug delivery with high encapsulation and 14 ± 2% release of curcumin into the supernatant in a week long study. Overall, the photocrosslinked CEC-D hydrogels exhibit stability, swelling ability, and sustained release of drugs.
[title size=”4″]Citation[/title]
Wang, Y.; Wang, X.; Montclare, J. K. Free-Standing Photocrosslinked Protein Polymer Hydrogels for Sustained Drug Release. Biomacromolecules 2021, 22 (4), 1509–1522. doi: 10.1021/acs.biomac.0c01721 [/toggle]
[toggle title=”From concept to reality: the use and impact of 3D prints as academic tools for high school biology education” open=”no”]
[title size=”3″]Abstract[/title]
While Science, Technology, Engineering, and Mathematics (STEM) education is essential in the high school curriculum, it often carries a reputation of being formidable and overwhelming. The resulting unease students often face inhibits their ability to learn effectively; thus it can be beneficial to integrate a more familiar approach to teach them such complex topics. We have developed a technology-enabled learning environment surrounding the use of 3D prints focused on the topics of homoeostasis and immunity for a ninth grade Living Environment class. The teacher and students’ surveys reveal that the 3D prints helped the students to effectively learn the material leading to a better understanding and retention of the topics. The results from the students’ performance, as well as the teacher and students’ feedback from the activity, demonstrate the effectiveness of 3D prints on maintaining their interest through a student-centred, student-led learning activity.
[title size=”4″]Citation[/title]
Monkovic, J. M.; Jones, S. M.; Nicolas, M.; Katyal, P.; Punia, K.; Noland, D.; Montclare, J. K. From concept to reality: the use and impact of 3D prints as academic tools for high school biology education. J. Biol. Educ. 2021, 1–13. doi: 10.1080/00219266.2020.1858927 [/toggle]
[toggle title=”Peptides as key components in the design of non‐viral vectors for gene delivery” open=”no”]
[title size=”3″]Abstract[/title]
Successful clinical implementation of gene delivery relies on the use of viral or non‐viral based vectors to package and protect the therapeutic nucleic acid. These vehicles must also be able to direct the fate of the cargo once it has entered the cell to ensure that the nucleic acid is functional, and the desired outcome is achieved. Compared to viral vectors, non‐viral vectors have the advantage of incorporating different material types such as lipids, polymers, and peptides to tune overall safety and efficacy. Peptides are especially powerful when used in gene delivery vectors as they are able to increase gene delivery efficacy by introducing new biochemical functionality. This review will discuss the use of peptides as central design components in non‐viral gene delivery vectors. The contribution of the peptide component to the overall functionality of the delivery vehicle will be highlighted, with a focus on peptides as the only vehicle component or peptides in complex assemblies with lipids or polymers.
[title size=”4″]Citation[/title]
Thomas, J.; Punia, K.; Montclare, J. K. Peptides as key components in the design of non-viral vectors for gene delivery. Peptide Sci. 2021, 113:e24189. doi: 10.1002/pep2.24189 [/toggle]
[toggle title=”Controlling Drug Absorption, Release, and Erosion of Photopatterned Protein Engineered Hydrogels” open=”no”]
[title size=”3″]Abstract[/title]
A protein-engineered triblock copolymer hydrogel composed of two self-assembling domains (SADs) has been fabricated by a photoactivatable diazirine group followed by ultraviolet (UV)-mediated crosslinking. The photocrosslinkable protein polymer CEC-D has been patterned into various features including different micrometer-scale stripes by using lithographic techniques. The patterned hydrogels are important for encapsulation of small molecules where a photopatterned fraction of 50% is optimal for maximum absorption. Stripe-patterned CEC-D100–100 exhibits slightly lower swelling ratios, an 8.9 times lower erosion profile, and a 2.6-fold higher drug release compared to the unpatterned hydrogel control, CEC-D0. Our studies demonstrate the potential of photocrosslinkable protein polymer hydrogels to be used as scaffolds for therapeutic delivery of small molecules. Through photolithographic techniques on the protein hydrogel, a variety of functionalities can be achieved by patterning different features enabling the mimicry of biological systems.
[title size=”4″]Citation[/title]
Wang, Y.; Delgado-Fukushima, E.; Fu, R. X.; Doerk, G. S.; Montclare, J. K. Controlling Drug Absorption, Release, and Erosion of Photopatterned Protein Engineered Hydrogels. Biomacromolecules 2020, 21 (9), 3608–3619. doi: 10.1021/acs.biomac.0c00616 [/toggle]
[toggle title=”Enhancing organophosphate hydrolase efficacy via protein engineering and immobilization strategies” open=”no”]
[title size=”3″]Abstract[/title]
Organophosphorus compounds (OPs), developed as pesticides and chemical warfare agents, are extremely toxic chemicals that pose a public health risk. Of the different detoxification strategies, organophosphate‐hydrolyzing enzymes have attracted much attention, providing a potential route for detoxifying those exposed to OPs. Phosphotriesterase (PTE), also known as organophosphate hydrolase (OPH), is one such enzyme that has been extensively studied as a catalytic bioscavenger. In this review, we will discuss the protein engineering of PTE aimed toward improving the activity and stability of the enzyme. In order to make enzyme utilization in OP detoxification more favorable, enzyme immobilization provides an effective means to increase enzyme activity and stability. Here, we present several such strategies that enhance the storage and operational stability of PTE/OPH.
[title size=”4″]Citation[/title] Katyal, P.; Chu, S.; Montclare, J. K. Enhancing organophosphate hydrolase efficacy via protein engineering and immobilization strategies. Ann. N.Y. Acad. Sci. 2020, 1480, 54–72. doi: 10.1111/nyas.14451 [/toggle]
[toggle title=”Recent trends in peptide and protein-based hydrogels” open=”no”]
[title size=”3″]Abstract[/title]
Hydrogels are classic examples of biomaterials that have found its niche in biomedical and allied fields. Here, we describe examples of peptide-based and protein-based hydrogels with a focus on smart gels that respond to various stimuli including temperature, pH, light, and ionic strength. With the recent advancements in computational modeling, it has been possible to predict as well as design peptide and protein sequences that can assemble into hydrogels with unique and improved properties. We briefly discuss coarse grained and atomistic simulations in designing peptides that can form hydrogels. In addition, we highlight the trends that will influence the future design and applications of hydrogels, with emphasis on bioadhesion, exosomes delivery, tissue and organoids engineering, and even intracellular production of gels.
[title size=”4″]Citation[/title]
Katyal, P.; Mahmoudinobar, F.; Montclare, J. K. Recent trends in peptide and protein-based hydrogels. Curr. Opin. Struct. Biol. 2020, 63, 97–105. doi: 10.1016/j.sbi.2020.04.007 [/toggle]
[/tab]
[tab id=2]
[accordian]
[toggle title=”Protein biomaterials for theranostic applications” open=”yes”]
[title size=”3″]Abstract[/title]
Protein biomaterials have been used for a wide range biomedical applications due to their intrinsic biocompatibility, versatility and higher order structure. Multifunctional theranostic agents fabricated by incorporating the diagnostic modalities and drug payloads in protein nanoformulations have garnered significant attention in recent years. Protein-based theranostic agents manifest high target specificity, enhanced blood circulation and reduce reticuloendothelial system elimination. This review focuses on the fabrication of peptide- and protein-based nanoformulations for imaging guided therapy.
[title size=”4″]Citation[/title]
Punia, K.; Kronenberg, J. B.; Montclare, J. K. Mol. Sys. Des. Eng. 2019, 4, 1074–1094. doi: 10.1039/C9ME00143C [/toggle]
[toggle title=”A ‘KAHOOT!’ Approach: The Effectiveness of Game-Based Learning for an Advanced Placement Biology Class” open=”no”]
[title size=”3″]Abstract[/title]
Keeping high school students engaged and motivated to learn complex scientific concepts can be difficult and challenging; this is especially true if the task feels daunting and unfamiliar to the students. Incorporating educational technology, such as KAHOOT, into the classroom can help students learn scientific material even when it is difficult. Our objective is to determine the effectiveness of gamification in an Advanced Placement Biology (AP biology) classroom by using the online game ‘KAHOOT!’ as a supplement to traditional teacher-centered learning. In addition, we determine the use of ‘KAHOOT!’ in enhancing student engagement and the learning experience for biology. A presentation on Transcription and Translation was given to a small group of high school AP Biology students (n = 18, 18 women). After the presentation, the students were given 15 questions and twenty seconds to answer each question in the ‘KAHOOT!’ game. Both the students and the teacher were given a post-activity survey to assess their interest in the activity. Based on the responses in the Students’ Survey, ‘KAHOOT!’ can be used as a gamified assessment tool to help students learn the topic of ‘Transcription and Translation’ by actively engaging them in a fun and exciting manner. The overall activity had a positive impact on the students and teacher as the students enjoyed learning Transcription and Translation through the use of ‘KAHOOT!’.
[title size=”4″]Citation[/title]
Jones, S. M.; Katyal, P.; Xie, X.; Nicolas, M. P.; Leung, E. M.; Noland, D. M.; Montclare, J. K. A ‘KAHOOT!’ Approach: The Effectiveness of Game-Based Learning for an Advanced Placement Biology Class. Simul. Gaming 2019, 50 (6), 832–847. doi:10.1177/1046878119882048 [/toggle]
[toggle title=”Thermoresponsive Protein-Engineered Coiled-Coil Hydrogel for Sustained Small Molecule Release” open=”no”]
[title size=”3″]Abstract[/title]
Thermoresponsive hydrogels are used for an array of biomedical applications. Lower critical solution temperature-type hydrogels have been observed in nature and extensively studied in comparison to upper critical solution temperature (UCST)-type hydrogels. Of the limited protein-based UCST-type hydrogels reported, none have been composed of a single coiled-coil domain. Here, we describe a biosynthesized homopentameric coiled-coil protein capable of demonstrating a UCST. Microscopy and structural analysis reveal that the hydrogel is stabilized by molecular entanglement of protein nanofibers, creating a porous matrix capable of binding the small hydrophobic molecule, curcumin. Curcumin binding increases the α-helical structure, fiber entanglement, mechanical integrity, and thermostability, resulting in sustained drug release at physiological temperature. This work provides the first example of a thermoresponsive hydrogel comprised of a single coiled-coil protein domain that can be used as a vehicle for sustained release and, by demonstrating UCST-type behavior, shows promise in forging a relationship between coiled-coil protein-phase behavior and that of synthetic polymer systems.
[title size=”4″]Citation[/title]
Hill, L. K.; Meleties, M.; Katyal, P. ; Xie, X.; Delgado-Fukushima, E.; Jihad, T.; Liu, C. -F.; O’Neill, S.; Tu, R. S.; Renfrew, P. D.; Bonneau, R.; Wadghiri, Y. Z.; Montclare, J. K. Thermoresponsive Protein-Engineered Coiled-Coil Hydrogel for Sustained Small Molecule Release. Biomacromolecules 2019, 20 (9), 3340–3351. doi: 10.1021/acs.biomac.9b00107 [/toggle]
[toggle title=”Self-Assembled Protein- and Peptide-Based Nanomaterials” open=”No”]
[title size=”3″]Abstract[/title]
Considerable effort has been devoted to generating novel protein- and peptide-based nanomaterials with their applications in a wide range of fields. Specifically, the unique property of proteins to self-assemble has been utilized to create a variety of nanoassemblies, which offer significant possibilities for next-generation biomaterials. In this minireview, we describe self-assembled protein- and peptide-based nanomaterials with focus on nanofibers and nanoparticles. Their applications in delivering therapeutic drugs and genes are discussed.
[title size=”4″]Citation[/title]
Katyal, P.; Meleties, M.; Montclare, J. K. Self-Assembled Protein- and Peptide-Based Nanomaterials. ACS Biomater. Sci. Eng. 2019, 5 (9), 4132–4147. doi: 10.1021/acsbiomaterials.9b00408 [/toggle]
[toggle title=”Protein‐Engineered Functional Materials” open=”no”]
[title size=”3″]Abstract[/title]
Proteins are versatile macromolecules that can perform a variety of functions. In the past three decades, they have been commonly used as building blocks to generate a range of biomaterials. Owing to their flexibility, proteins can either be used alone or in combination with other functional molecules. Advances in synthetic and chemical biology have enabled new protein fusions as well as the integration of new functional groups leading to biomaterials with emergent properties. This review discusses protein‐engineered materials from the perspectives of domain‐based designs as well as physical and chemical approaches for crosslinked materials, with special emphasis on the creation of hydrogels. Engineered proteins that organize or template metal ions, bear noncanonical amino acids (NCAAs), and their potential applications, are also reviewed.
[title size=”4″]Citation[/title]
Wang, Y.; Katyal, P.; Montclare, J. K. Protein-Engineered Functional Materials. Adv. Healthc. Mater. 2019, 8, 1801374. doi: 10.1002/adhm.201801374 [/toggle]
[toggle title=”Protein-Engineered Nanoscale Micelles for Dynamic ¹⁹F Magnetic Resonance and Therapeutic Drug Delivery” open=”no”]
[title size=”3″]Abstract[/title]
Engineered proteins provide an interesting template for designing fluorine-19 (¹⁹F) magnetic resonance imaging (MRI) contrast agents, yet progress has been hindered by the unpredictable relaxation properties of fluorine. Herein, we present the biosynthesis of a protein block copolymer, termed “fluorinated thermoresponsive assembled protein” (F-TRAP), which assembles into a monodisperse nanoscale micelle with interesting ¹⁹F NMR properties and the ability to encapsulate and release small therapeutic molecules, imparting potential as a diagnostic and therapeutic (theranostic) agent. The assembly of the F-TRAP micelle, composed of a coiled-coil pentamer corona and a hydrophobic, thermoresponsive elastin-like polypeptide core, results in a drastic depression in spin–spin relaxation (T2) times and unaffected spin–lattice relaxation (T1) times. The nearly unchanging T1 relaxation rates and linearly dependent T2 relaxation rates have allowed for detection via zero echo time ¹⁹F MRI, and the in vivo MR potential has been preliminarily explored using ¹⁹F magnetic resonance spectroscopy (MRS). This fluorinated micelle has also demonstrated the ability to encapsulate the small-molecule chemotherapeutic doxorubicin and release its cargo in a thermoresponsive manner owing to its inherent stimuli-responsive properties, presenting an interesting avenue for the development of thermoresponsive ¹⁹F MRI/MRS-traceable theranostic agents.
[title size=”4″]Citation[/title]
Hill, L. K.; Frezzo, J. A.; Katyal, P.; Hoang, D. H.; Gironda, Z. B. Y.; Xu, C.; Xie, X.; Delgado-Fukushima, E.; Wadghiri, Y. Z.; Montclare, J. K. Protein-Engineered Nanoscale Micelles for Dynamic ¹⁹F Magnetic Resonance and Therapeutic Drug Delivery. ACS Nano. 2019, 13 (3), 2969–2985. doi: 10.1021/acsnano.8b07481 [/toggle]
[toggle title=”Efficient siRNA delivery and gene silencing using a lipopolypeptide hybrid vector mediated by a caveolae-mediated and temperature-dependent endocytic pathway” open=”no”]
[title size=”3″]Abstract[/title]
We developed a non-viral vector, a combination of HIV-1 Tat peptide modified with histidine and cysteine (mTat) and polyethylenimine, jetPEI (PEI), displaying the high efficiency of plasmid DNA transfection with little toxicity. Since the highest efficiency of INTERFERin (INT), a cationic amphiphilic lipid-based reagent, for small interfering RNA (siRNA) transfection among six commercial reagents was shown, we hypothesized that combining mTat/PEI with INT would improve transfection efficiency of siRNA delivery. To elucidate the efficacy of the hybrid vector for siRNA silencing, β-actin expression was measured after siRNA β-actin was transfected with mTat/PEI/INT or other vectors in HSC-3 human oral squamous carcinoma cells.
[title size=”4″]Citation[/title]
Kasai, H.; Inoue, K. Imamura, K. Yuvienco, C.; Montclare, J. K.; Yamano, S. J.Nanobiotechnology 2019, 17 (1), 11. doi: 10.1186/s12951-019-0444-8 [/toggle]
[toggle title=”Engineered Coiled-Coil Protein for Delivery of Inverse Agonist for Osteoarthritis” open=”No”]
[title size=”3″]Abstract[/title]
Recombinant methods have been used to engineer artificial protein triblock polymers composed of two different self-assembling domains (SADs) bearing one elastin (E) flanked by two cartilage oligomeric matrix protein coiled-coil (C) domains to generate CEC. To understand how the two C domains improve small molecule recognition and the mechanical integrity of CEC, we have constructed CL44AECL44A, which bears an impaired CL44A domain that is unstructured as a negative control. The CEC triblock polymer demonstrates increased small molecule binding and ideal elastic behavior for hydrogel formation. The negative control CL44AECL44A does not exhibit binding to small molecule and is inelastic at lower temperatures, affirming the favorable role of C domain and its helical conformation. While both CEC and CL44AECL44A assemble into micelles, CEC is more densely packed with C domains on the surface enabling the development of networks leading to hydrogel formation. Such protein engineered triblock copolymers capable of forming robust hydrogels hold tremendous promise for biomedical applications in drug delivery and tissue engineering.
[title size=”4″]Citation[/title]
Yin, L.; Agustinus, A. S.; Yuvienco, C.; Minashima, T.; Schnabel, N. L.; Kirsch, T.; Montclare, J. K. Engineered Coiled-Coil Protein for Delivery of Inverse Agonist for Osteoarthritis. Biomacromolecules 2018, 19 (5), 1614–1624. doi: 10.1021/acs.biomac.8b00158 [/toggle]
[toggle title=”Protein Engineered Triblock Polymers Composed of Two SADs: Enhanced Mechanical Properties and Binding Abilities” open=”No”]
[title size=”3″]Abstract[/title]
Recombinant methods have been used to engineer artificial protein triblock polymers composed of two different self-assembling domains (SADs) bearing one elastin (E) flanked by two cartilage oligomeric matrix protein coiled-coil (C) domains to generate CEC. To understand how the two C domains improve small molecule recognition and the mechanical integrity of CEC, we have constructed CL44AECL44A, which bears an impaired CL44A domain that is unstructured as a negative control. The CEC triblock polymer demonstrates increased small molecule binding and ideal elastic behavior for hydrogel formation. The negative control CL44AECL44A does not exhibit binding to small molecule and is inelastic at lower temperatures, affirming the favorable role of C domain and its helical conformation. While both CEC and CL44AECL44A assemble into micelles, CEC is more densely packed with C domains on the surface enabling the development of networks leading to hydrogel formation. Such protein engineered triblock copolymers capable of forming robust hydrogels hold tremendous promise for biomedical applications in drug delivery and tissue engineering.
[title size=”4″]Citation[/title]
Olsen, A. J.; Katyal, P.; Haghpanah, J. S.; Kubilius, M. B.; Li, R.; Schnabel, N. L.; O’Neill, S. C.; Wang, Y.; Dai, M.; Singh, N.; Tu, R. S.; Montclare, J. K. Protein Engineered Triblock Polymers Composed of Two SADs: Enhanced Mechanical Properties and Binding Abilities. Biomacromolecules 2018, 19 (5), 1552–1561. doi: https://doi.org/10.1021/acs.biomac.7b01259 [/toggle]
[toggle title=”Family Matters: How Family Influenced My Career” open=”No”]
[title size=”3″]Abstract[/title]
My family emigrated from Korea, and I was born and raised in the Bronx. I was taught to value education, and my parents worked several jobs just so my brother and I could go to good schools. I loved math and science all throughout my life and got to experience research during my high school career. My love was fostered by my teachers, and, while I never considered an academic scientist as a career, it was my teachers and mentors who led me on my path.
[title size=”4″]Citation[/title]
Montclare, J. K. Family Matters: How Family Influenced My Career. In Mom the Chemistry Professor; Woznack, K. Charlebois, A., Cole R., Marzabadi C., Webster G., Eds.; Springer: Cham, Germany, 2018; pp 345–353. doi: 10.1007/978-3-319-78972-9_25 [/toggle]
[toggle title=”Design and Characterization of Fibers and Bionanocomposites Using the Coiled-Coil Domain of Cartilage Oligomeric Matrix Protein” open=”No”]
[title size=”3″]Abstract[/title]
Tremendous effort has been dedicated to the design and assembly of bioinspired protein-based architectures with potential applications in drug delivery, tissue engineering, biosensing, and bioimaging. Here, we describe our strategy to generate fibers and bionanocomposites using the coiled-coil domain of cartilage oligomeric matrix protein (COMPcc). Our construct, Q, engineered by swapping particular regions of COMPcc to optimize surface charge, self-assembles to form nanofibers. The Q protein nanofibers can efficiently bind curcumin to form robust mesofibers that can be potentially used for drug delivery and biomedical applications. In addition, using the same Q protein, we describe the biotemplation of gold nanoparticles (AuNP) in the presence and absence of the hexahistidine tag (His-tag). The Q bearing His-tag·AuNP (Q·AuNP) readily deposits on electrode surfaces, while Q without His-tag·AuNP (Qx·AuNP) stabilizes the soluble protein·gold bionanocomposites for several days without aggregating.
[title size=”4″]Citation[/title]
Katyal, P. and Montclare, J. K. Design and characterization of fibers and bionanocomposites using the coiled-coil domain of cartilage oligomeric matrix protein. In Protein Scaffolds. Methods in Molecular Biology, vol 1798; Humana Press: New York, NY, USA, 2018; pp. 239–263. doi: 10.1007/978-1-4939-7893-9_19 [/toggle]
[toggle title=”Perspectives on Blended Learning through the On-Line Platform, LabLessons, for Chemistry” open=”No”]
[title size=”3″]Abstract[/title]
The effectiveness of blended learning was evaluated through the integration of an online chemistry platform, LabLessons. Two modules, Formation of Hydrogen and Titration, were designed by college mentors alongside classroom chemistry teachers to engage and allow high school students to better comprehend these scientific topics. The pre-lab modules introduced the students to experiments they were expected to perform in class the following day. The modules consisted of an introduction as well as either a visualization and/or simulation specific to each topic. Students and teachers who utilized LabLessons were surveyed to establish a preliminary research on the use of technology in classrooms. Student and teacher surveys demonstrated LabLessons to be an interactive and helpful tool to improve students’ understanding of conceptual ideas.
[title size=”4″]Citation[/title]
Jihad, T.; Klementowicz, E.; Gryczka, P.; Sharrock, C.; Maxfield, M.; Lee, Y.; Montclare, J. K. Perspectives on blended learning through the on-line platform, lablessons, for chemistry. J. Technol. Sci. Educ. 2018, 8 (1), 34–44. doi: 10.3926/jotse.312 [/toggle]
[toggle title=”Efficient Dual siRNA and Drug Delivery Using Engineered Lipoproteoplexes” open=”no”]
[title size=”3″]Abstract[/title]
An engineered supercharged coiled-coil protein (CSP) and the cationic transfection reagent Lipofectamine 2000 are combined to form a lipoproteoplex for the purpose of dual delivery of siRNA and doxorubicin. CSP, bearing an external positive charge and axial hydrophobic pore, demonstrates the ability to condense siRNA and encapsulate the small-molecule chemotherapeutic, doxorubicin. The lipoproteoplex demonstrates improved doxorubicin loading relative to Lipofectamine 2000. Furthermore, it induces effective transfection of GAPDH (60% knockdown) in MCF-7 breast cancer cells with efficiencies comparing favorably to Lipofectamine 2000. When the lipoproteoplex is loaded with doxorubicin, the improved doxorubicin loading (∼40 μg Dox/mg CSP) results in a substantial decrease in MCF-7 cell viability.
[title size=”4″]Citation[/title]
Liu, C. F.; Chen, R.; Frezzo, J. A.; Katyal, P.; Hill, L. K.; Yin, L.; Srivastava, N.; More, H. T.; Renfrew, P. D.; Bonneau, R.; Montclare, J. K. Efficient Dual siRNA and Drug Delivery Using Engineered Lipoproteoplexes. Biomacromolecules 2017, 18 (9), 2688–2698. doi: 10.1021/acs.biomac.7b00203 [/toggle]
[toggle title=”Protein based therapeutic delivery agents: Contemporary developments and challenges” open=”no”]
[title size=”3″]Abstract[/title]
As unique biopolymers, proteins can be employed for therapeutic delivery. They bear important features such as bioavailability, biocompatibility, and biodegradability with low toxicity serving as a platform for delivery of various small molecule therapeutics, gene therapies, protein biologics and cells. Depending on size and characteristic of the therapeutic, a variety of natural and engineered proteins or peptides have been developed. This, coupled to recent advances in synthetic and chemical biology, has led to the creation of tailor-made protein materials for delivery. This review highlights strategies employing proteins to facilitate the delivery of therapeutic matter, addressing the challenges for small molecule, gene, protein and cell transport.
[title size=”4″]Citation[/title]
Yin, L.; Yuvienco, C.; Montclare, J. K. Protein based therapeutic delivery agents: Contemporary developments and challenges. Biomaterials 2017, 134, 91–116. doi: 10.1016/j.biomaterials.2017.04.036 [/toggle]
[toggle title=”Novel lipoproteoplex delivers Keap1 siRNA based gene therapy to accelerate diabetic wound healing.” open=”no”]
[title size=”3″]Abstract[/title]
Therapeutics utilizing siRNA are currently limited by the availability of safe and effective delivery systems. Cutaneous diseases, specifically ones with significant genetic components are ideal candidates for topical siRNA based therapy but the anatomical structure of skin presents a considerable hurdle. Here, we optimized a novel liposome and protein hybrid nanoparticle delivery system for the topical treatment of diabetic wounds with severe oxidative stress. We utilized a cationic lipid nanoparticle (CLN) composed of 1,2-dioleoyl-3-trimethylammonium-propane (DOTAP) and the edge activator sodium cholate (NaChol), in a 6:1 ratio of DOTAP:NaChol (DNC). Addition of a cationic engineered supercharged coiled-coil protein (CSP) in a 10:1:1 ratio of DNC:CSP:siRNA produced a stable lipoproteoplex (LPP) nanoparticle, with optimal siRNA complexation, minimal cytotoxicity, and increased transfection efficacy. In a humanized murine diabetic wound healing model, our optimized LPP formulation successfully delivered siRNA targeted against Keap1, key repressor of Nrf2 which is a central regulator of redox mechanisms. Application of LPP complexing siKeap1 restored Nrf2 antioxidant function, accelerated diabetic tissue regeneration, and augmented reduction-oxidation homeostasis in the wound environment. Our topical LPP delivery system can readily be translated into clinical use for the treatment of diabetic wounds and can be extended to other cutaneous diseases with genetic components.
[title size=”4″]Citation[/title]
Rabbani, P. S.; Zhou, A.; Borab, Z. M.; Frezzo, J. A.; Srivastava, N.; More, H. T.; Rifkin, W. J.; David, J. A.; Berens, S. J.; Chen, R.; Hameedi, S.; Junejo, M. H.; Kim, C.; Sartor, R. A.; Liu, C. F.; Saadeh, P. B.; Montclare, J. K.; Ceradini, D. J. Novel lipoproteoplex delivers Keap1 siRNA based gene therapy to accelerate diabetic wound healing. Biomaterials 2017, 132, 1–15. doi: 10.1016/j.biomaterials.2017.04.001 [/toggle]
[toggle title=”Impact of phenylalanines outside the dimer interface on phosphotriesterase stability and function” open=”no”]
[title size=”3″]Abstract[/title]
We explore the significance of phenylalanine outside of the phosphotriesterase (PTE) dimer interface through mutagenesis studies and computational modeling. Previous studies have demonstrated that the residue-specific incorporation of para-fluorophenylalanine (pFF) into PTE improves stability, suggesting the importance of phenylalanines in stabilization of the dimer. However, this comes at a cost of decreased solubility due to pFF incorporation into other parts of the protein. Motivated by this, eight single solvent-exposed phenylalanine mutants are evaluated via ROSETTA and good correspondence between experiments and these predictions is observed. Three residues, F304, F327, and F335, appear to be important for PTE activity and stability, even though they do not reside in the dimer interface region or active site. While the remaining mutants do not significantly affect structure or activity, one variant, F306L, reveals improved activity at ambient and elevated temperatures. These studies provide further insight into role of these residues on PTE function and stability.
[title size=”4″]Citation[/title]
Olsen, A. J.; Halvorsen, L. A.; Yang, C. -Y.; Ventura, R. B.; Yin, L.; Renfrew, P. D.; Bonneau, R.; Montclare, J. K. Impact of phenylalanines outside the dimer interface on phosphotriesterase stability and function. Mol. BioSyst. 2017, 13, 2092–2106. doi: 10.1039/c7mb00196g [/toggle]
[toggle title=”Natural Composite Systems for Bioinspired Materials” open=”no”]
[title size=”3″]Abstract[/title]
From a relatively limited selection of base materials, nature has steered the development of truly remarkable materials. The simplest and often overlooked organisms have demonstrated the ability to manufacture multi-faceted, molecular-level hierarchical structures that combine mechanical properties rarely seen in synthetic materials. Indeed, these natural composite systems, composed of an array of intricately arranged and functionally relevant organic and inorganic substances serve as inspiration for materials design. A better understanding of these composite systems, specifically at the interface of the hetero-assemblies, would encourage faster development of environmentally friendly “green” materials with molecular level specificities.
[title size=”4″]Citation[/title]
Frezzo, J. A. and Montclare, J. K. Natural Composite Systems for Bioinspired Materials. In Protein-based Engineered Nanostructures. Advances in Experimental Medicine and Biology, vol 940; Cortajarena, A., Grove, T. Eds.; Springer: Cham, Germany, 2016; pp 143–166. doi: 10.1007/978-3-319-39196-0_7 [/toggle]
[toggle title=”LabLessons: Effects of Electronic Prelabs on Student Engagement and Performance” open=”no”]
[title size=”3″]Abstract[/title]
Lab instructors, for both high school and undergraduate college level courses, face issues of constricted time within the lab period and limited student engagement with prelab materials. To address these issues, an online prelab delivery system named LabLessons is developed and tested out in a high school chemistry classroom. The system supplements the laboratory experience by providing visualizations and simulations of concepts to prepare students for the practical experiments. The system requires students to answer prelab questions online, which provides immediate feedback and cuts down on last minute copying of answers that instructors anecdotally reported with paper laboratories. Empirical results demonstrate the effectiveness and improved outcomes for students who have used LabLessons. In addition, the ease of use of the system and better preparedness for the lab is noted by the instructor.
[title size=”4″]Citation[/title]
Gryczka, P.; Klementowicz, E.; Sharrock, C.; Maxfield, M.; Montclare, J. K. LabLessons: Effects of Electronic Prelabs on Student Engagement and Performance. J. Chem. Educ. 2016, 93 (12), 2012–2017. doi: 10.1021/acs.jchemed.6b00394 [/toggle]
[toggle title=”Interactive Online Physics Labs Increase High School Students’ Interest” open=”no”]
[title size=”3″]Abstract[/title]
Here we describe the incorporation of a web-based application focusing on circuits for the physics high school classroom as part of an outreach program. The program involves college mentors creating and implementing science lessons in collaboration with the classroom teacher. Focusing on the challenge of understanding circuit design, a technology rich module is employed to improve learning and motivation of the students. The students’ conceptual understanding as well as interest in circuits was increased, the college mentors earned valuable teaching and mentoring experience and the teacher enjoyed more one-on-one time as well as assistance with students.
[title size=”4″]Citation[/title]
Gryczka, P.; Klementowicz, E.; Sharrock, C.; Montclare, J. Interactive online physics labs increase high school students’ interest. J. Technol. Sci. Educ. 2016, 6 (3), 166–187. doi: 10.3926/jotse.191 [/toggle]
[toggle title=”Engineered Protein Polymer-Gold Nanoparticle Hybrid Materials for Small Molecule Delivery” open=”no”]
[title size=”3″]Abstract[/title]
We have fabricated protein polymer-gold nanoparticle (P-GNP) nanocomposites that exhibit enhanced binding and delivery properties of the small hydrophobic molecule drug, curcumin, to the model breast cancer cell line, MCF-7. These hybrid biomaterials are constructed via in situ GNP templated-synthesis with genetically engineered histidine tags. The P-GNP nanocomposites exhibit enhanced small molecule loading, sustained release and increased uptake by MCF-7 cells. When compared to the proteins polymers alone, the P-GNPs demonstrated a greater than 7-fold increase in curcumin binding, a nearly 50% slower release profile and more than 2-fold increase in cellular uptake of curcumin. These results suggest that P-GNP nanocomposites serve as promising candidates for drug delivery vehicles
[title size=”4″]Citation[/title]
Dai, M.; Frezzo, J. A. Sharma, E.; Chen, R.; Singh, N.; Yuvienco, C.; Caglar, E.; Xiao, S.; Saxena, A.; Montclare, J. K. Engineered Protein Polymer-Gold Nanoparticle Hybrid Materials for Small Molecule Delivery. J. Nanomed. Nanotechnol. 2016, 7 (1), 356. doi: 10.4172/2157-7439.1000356 [/toggle]
[toggle title=”Exploring the potential of engineered coiled-coil protein microfibers in drug delivery” open=”no”]
[title size=”3″]Editorial[/title]
Over the course of evolution, nature has made extensive use of the α-helical coiled-coil for a wide breadth of applications in gene regulation, mitosis, cellular and muscular motility as well as cytoskeletal structure. An estimated 2-10% of all proteins harbor the coiled-coil motif, demonstrating the versatility of this particular motif across the spectrum of protein functions. While the functional diversity of the coiled-coil motif in nature is well established, there are expanding roles these motifs may play in tissue engineering and drug delivery. Before delving into this promising field of coiled-coil proteins as drug delivery vehicles, it is pertinent to discuss the underlying principles that govern the assembly of the coiled-coil.
[title size=”4″]Citation[/title]
Frezzo, J. A. and Montclare, J. K. Exploring the potential of engineered coiled-coil protein microfibers in drug delivery. Ther. Deliv. 2015, 6 (6), 643–646. doi: 10.4155/tde.15.19 [/toggle]
[toggle title=”Tunable Conformation-Dependent Engineered Protein·Gold Nanoparticle Nanocomposites” open=”no”]
[title size=”3″]Abstract[/title]
We demonstrate the fabrication of protein·gold nanoparticle (AuNP) nanocomposites in situ, leading to distinct assemblies dependent upon protein secondary structure. In the presence of pentameric coiled-coil proteins C and Q, which contain histidine tags and have helicities of 54 and 65%, respectively, templation of AuNP results in precipitation of the protein· AuNP composites with AuNPs 6.5 nm in diameter, creating macromolecular assemblies on the micrometer scale. In the absence of the histidine tags, the resulting Cx and Qx proteins, which exhibit lower helicities of 37 and 45%, respectively, stabilize soluble protein·AuNP composites with AuNPs 4.5 nm in diameter for several days without aggregating. By manipulating protein structure via external triggers, such as TFE, we obtain control over the macromolecular conformation and overall physicochemical properties. These hybrid protein·AuNP assemblies can be readily deposited on electrodes, where they can serve as a tunable bionanocomposite kinetic barrier.
[title size=”4″]Citation[/title]
Hume, J.; Chen, R.; Jacquet, R.; Yang, M.; Montclare, J. K. Tunable Conformation-Dependent Engineered Protein·Gold Nanoparticle Nanocomposites. Biomacromolecules 2015, 16 (6), 1706–1713. doi: 10.1021/acs.biomac.5b00098 [/toggle]
[toggle title=”Influence of Fluorination on Protein Engineered Coiled-coil Fibers” open=”no”]
[title size=”3″]Abstract[/title]
We describe the design and characterization of fluorinated coiled-coil proteins able to assemble into robust nanoand microfibers. Fluorination is achieved biosynthetically by residue-specific incorporation of 5,5,5-trifluoroleucine (TFL). The fluorinated proteins C+TFL and Q+TFL are highly α-helical as confirmed via circular dichroism (CD) and more resistant to thermal denaturation compared to their nonfluorinated counterparts, C and Q. The fluorinated proteins demonstrate enhanced fiber assembly at pH 8.0 with higher order structure in contrast to nonfluorinated proteins, which are unable to form fibers under the same conditions. Ionic strength dependent fiber assembly is observed for fluorinated as well as wild-type proteins in which the fluorinated proteins exhibited more stable, thicker fibers. The fluorinated and nonfluorinated proteins reveal metal ion-dependent small molecule recognition and supramolecular assemblies. In the presence of Zn (II), enhanced thermal stability and fiber assembly is observed for the fluorinated proteins and their nonfluorinated counterparts. Whereas Ni (II) promotes aggregation with no fiber assembly, the stabilization of α-helix by Zn (II) results in enhanced binding to curcumin by the fluorinated proteins. Surprisingly, the nonfluorinated proteins exhibit multiplefold increase in curcumin binding in the presence of Zn (II). In the context of the growing number of protein-based fiber assemblies, these fluorinated coiled-coil proteins introduce a new paradigm in the development of highly stable, robust selfassembling fibers under more physiologically relevant pH conditions that promotes the binding and release of small molecules in response to external cues.
[title size=”4″]Citation[/title]
More, H. T.; Zhang, K. S.; Srivastava, N.; Frezzo, J. A.; Montclare, J. K. Influence of Fluorination on Protein-Engineered Coiled-Coil Fibers. Biomacromolecules 2015, 16 (4), 1210–1217. doi: 10.1021/bm5019062 [/toggle]
[toggle title=”Integrating Technology in STEM Education” open=”no”]
[title size=”3″]Abstract[/title]
Students have access to the Internet at their fingertips via e-tablets and smart phones. However, the STEM fields are struggling to remain relevant in students’ lives outside the classroom. In an effort to improve high school science curricula and to keep students engaged in the classroom, we developed a technology-rich bioengineering summer program for high school students in grades 9-12. The program utilized touch screen technology in conjunction with hands-on experiments and traditional lecturing to create an entertaining, relevant, and effective classroom experience.
[title size=”4″]Citation[/title]
Chacko, P.; Appelbaum, S.; Kim, H.; Zhao, J.; Montclare, J. K. Integrating Technology in STEM Education. J. Technol. Sci. Educ. 2015, 5 (1), 5–14. doi: 10.3926/jotse.124 [/toggle]
[toggle title=”Using Touch-Screen Technology, Apps, and Blogs to Engage and Sustain High School Students’ Interest in Chemistry Topics” open=”no”]
[title size=”3″]Abstract[/title]
As part of an outreach program, we integrated chemistry apps with blogging to enhance the learning experience of students in and outside the classroom. Our outreach program involved college mentors who participated in the development and implementation of chemistry lessons alongside the classroom teacher. Three technology-rich modules that focused on molecules, balancing equations, and nuclear chemistry were taught to high school students. Feedback-oriented and interactive lessons through apps with blogging were used to engage the students with the instructors as well as their peers. The combination of blogging with interactive apps increased student involvement and sustained their interests in the chemistry topics covered by the modules. The students were divided into experimental and control groups. The experimental groups were required to use the blog, where students viewed the questions and uploaded their answers and comments. The control group did not have access to the blog during the modules. Performance, class participation, and interest in STEM fields by the groups were examined. The students received a personalized, interactive learning experience in chemistry, the college mentors gained teaching and mentoring experience, and the teacher received assistance in implementing technology in the classroom.
[title size=”4″]Citation[/title]
Kim, H.; Chacko, P.; Zhao, J.; Montclare, J. K. Using Touch-Screen Technology, Apps, and Blogs To Engage and Sustain High School Students’ Interest in Chemistry Topics. J. Chem. Educ. 2014, 91 (11), 1818–1822. doi: 10.1021/ed500234z [/toggle]
[toggle title=”Engineered Coiled-Coil Protein Microfibers” open=”no”]
[title size=”3″]Abstract[/title]
The fabrication of de novo proteins able to self-assemble on the nano- to meso-length scales is critical in the development of protein-based biomaterials in nanotechnology and medicine. Here we report the design and characterization of a protein engineered coiled-coil that not only assembles into microfibers, but also can bind hydrophobic small molecules. Under ambient conditions, the protein forms fibers with nanoscale structure possessing large aspect ratios formed by bundles of α-helical homopentameric assemblies, which further assemble into mesoscale fibers in the presence of curcumin through aggregation. Surprisingly, these biosynthesized fibers are able to form in conditions of remarkably low concentrations. Unlike previously designed coiled-coil fibers, these engineered protein microfibers can bind the small molecule curcumin throughout the assembly, serving as a depot for encapsulation and delivery of other chemical agents within protein-based 3D microenvironments.
*highlighted in https://www.fiercepharma.com/drug-delivery/nyu-researchers-break-nano-barrier-to-engineer-first-protein-microfiber
[title size=”4″]Citation[/title]
Hume, J.; Sun, J.; Jacquet, R.; Renfrew, P. D.; Martin, J. A.; Bonneau, R.; Gilchrist, M. L.; Montclare, J. K. Engineered Coiled-Coil Protein Microfibers. Biomacromolecules 2014, 15 (10), 3503–3510. doi: 10.1021/bm5004948 [/toggle]
[toggle title=”Improved Stability and Half-life of Fluorinated Phosphotriesterase using Rosetta” open=”no”]
[title size=”3″]Abstract[/title]
Recently we demonstrated that incorporating p-fluorophenylalanine (pFF) into phosphotriesterase dramatically improved folding, thereby leading to enhanced stability and function at elevated temperatures. To further improve the stability of the fluorinated enzyme, Rosetta was used to identify multiple potential stabilizing mutations. One such variant, pFF-F104A, exhibited enhanced activity at elevated temperature and maintained activity over many days in solution at room temperature.
*highlighted in https://www.medicalnewstoday.com/articles/280450
[title size=”4″]Citation[/title]
Yang, C. -Y.; Renfrew, P. D.; Olsen, A. J.; Zhang, M.; Yuvienco, C.; Bonneau, R.; Montclare, J. K. Improved Stability and Half-Life of Fluorinated Phosphotriesterase Using Rosetta. ChemBioChem 2014, 15 (12), 1761–1764. doi: 10.1002/cbic.201402062 [/toggle]
[toggle title=”Gene delivery from supercharged coiled-coil protein and cationic lipid hybrid complex” open=”no”]
[title size=”3″]Abstract[/title]
A lipoproteoplex comprised of an engineered supercharged coiled-coil protein (CSP) bearing multiple arginines and the cationic lipid formulation FuGENE HD (FG) was developed for effective condensation and delivery of nucleic acids. The CSP was able to maintain helical structure and self-assembly properties while exhibiting binding to plasmid DNA. The ternary CSP·DNA(8:1)·FG lipoproteoplex complex demonstrated enhanced transfection of β-galactosidase DNA into MC3T3-E1 mouse preosteoblasts. The lipoproteoplexes showed significant increases in transfection efficiency when compared to conventional FG and an mTat·FG lipopolyplex with a 6- and 2.5-fold increase in transfection, respectively. The CSP·DNA(8:1)·FG lipoproteoplex assembled into spherical particles with a net positive surface charge, enabling efficient gene delivery. These results support the application of lipoproteoplexes with protein engineered CSP for non-viral gene delivery.
[title size=”4″]Citation[/title]
More, H. T.; Frezzo, J. A.; Dai, J.; Yamano, S.; Montclare, J. K. Gene delivery from supercharged coiled-coil protein and cationic lipid hybrid complex. Biomaterials 2014, 35 (25), 7188–7193. doi: 10.1016/j.biomaterials.2014.05.005 [/toggle]
[toggle title=”Long-term efficient gene delivery using polyethylenimine with modified Tat peptide” open=”no”]
[title size=”3″]Abstract[/title]
Polyethylenimine (PEI), a cationic polymer, has been widely studied and shown great promise as an efficient gene delivery vehicle. Likewise, the HIV-1 Tat peptide, a cell-permeable peptide, has been successfully used for intracellular gene delivery. To improve the favorable properties of these two vectors, we combine PEI with the modified Tat peptide sequence bearing histidine and cysteine residues (mTat). In vitro mTat/PEI-mediated transfection was evaluated by luciferase expression plasmid in two cell types. mTat/PEI produced significant improvement (≈5-fold) in transfection efficiency of both cell lines with little cytotoxicity when compared to mTat alone, PEI alone, or four commercial reagents. The particle size of mTat/PEI/DNA complex was significantly smaller than mTat or PEI alone, and it was correlated with higher transfection efficiency. Filipin III, an inhibitor of caveolae-mediated endocytosis, significantly inhibited mTat/PEI transfection. In contrast, chlorpromazine, an inhibitor of clathrin-mediated endocytosis, did not. This suggested caveolae-mediated endocytosis as the transfection mechanism. Furthermore, the results of in vivo studies showed that animals administered mTat/PEI/DNA intramuscularly had significantly higher and longer luciferase expression (≈7 months) than those with mTat/DNA, PEI/DNA, or DNA alone, without any associated toxicity. The combination of mTat with PEI could significantly improve transfection efficiency, expanding the potential use as a non-viral gene vector both in vitro and in vivo.
[title size=”4″]Citation[/title]
Yamano, S.; Dai, J.; Hanatani, S.; Haku, K.; Yamanaka, T.; Ishioka, M.; Takayama, T.; Yuvienco, C.; Khapli, S.; Moursi, A. M.; Montclare, J. K. Biomaterials 2014, 35 (5),1705–1715. doi: 10.1016/j.biomaterials.2013.11.012 [/toggle]
[toggle title=”Bionanocomposites: Differential Effects of Cellulose Nanocrystals on Protein Diblock Copolymers” open=”no”]
[title size=”3″]Abstract[/title]
We investigate the effects of mixing a colloidal suspension of tunicate-derived cellulose nanocrystals (t-CNCs) with aqueous colloidal suspensions of two protein diblock copolymers, EC and CE, which bear two different self-assembling domains (SADs) derived from elastin (E) and the coiled-coil region of cartilage oligomeric matrix protein (C). The resulting aqueous mixtures reveal improved mechanical integrity for the CE+t-CNC mixture, which exhibits an elastic gel network. This is in contrast to EC+t-CNC, which does not form a gel, indicating that block orientation influences the ability to interact with t-CNCs. Surface analysis and interfacial characterization indicate that the differential mechanical properties of the two samples are due to the prevalent display of the E domain by CE, which interacts more with t-CNCs leading to a stronger network with t-CNCs. On the other hand, EC, which is predominantly C-rich on its surface, does not interact as much with t-CNCs. This suggests that the surface characteristics of the protein polymers, due to folding and self-assembly, are important factors for the interactions with t-CNCs, and a significant influence on the overall mechanical properties. These results have interesting implications for the understanding of natural composite biomaterials and the development of artificially assembled bionanocomposites.
[title size=”4″]Citation[/title]
[cite]10.1021/bm401304w[/cite][/toggle]
[toggle title=”Increased osseointegration effect of bone morphogenetic protein 2 on dental implants: An in vivo study” open=”no”]
[title size=”3″]Abstract[/title]
Application of recombinant human bone morphogenetic protein 2 (rhBMP-2) to implant surfaces has been of great interest due to its osteoinductive potential. However, the optimal coating methodology has not been clarified. The objective of the study was to determine whether the application of rhBMP-2 onto plasma-sprayed hydroxyapatite implant surfaces by immersion in protein solution before implant installation would result in significantly improved bone apposition. Using a sheep iliac model, titanium (Ti) and plasma-sprayed calcium–phosphate (PSCaP)-coated implants uncoated and coated with rhBMP-2 were assessed for their osteogenic effects in the peri-implant area over time in terms of osseointegration and de novo bone formation. After 3 and 6 weeks postoperatively, the samples were retrieved and were subjected to bone-to-implant contact (BIC) and bone area fraction occupancy (BAFO) evaluation. When rhBMP-2 was applied to the PSCaP surface, significant increases in BIC and BAFO were observed at 3 weeks in vivo, whereas when adsorbed directly onto the titanium implant surface, rhBMP-2 did not as effectively improve the bone response (although significantly higher than control Ti). The outcomes of the present study suggested that the combination of plasma-sprayed calcium–phosphate surface and rhBMP-2 coating significantly enhanced osseointegration, which validated the postulated hypothesis.
[title size=”4″]Citation[/title]
Yoo, D.; Tover, N.; Jimbo, R.; Marin, C.; Anchieta, R. B.; Machado, L. S.; Montclare, J.; Guastaldi, F. P. S.; Janal, M. N.; Coelho, P. G. Increased osseointegration effect of bone morphogenetic protein 2 on dental implants: An in vivo study. J. Biomed. Mater. Res. 2014, 102 (6), 1921–1927. doi: 10.1002/jbm.a.34862 [/toggle]
[toggle title=”Force-Clamp Experiments Reveal the Free-Energy Profile and Diffusion Coefficient of the Collapse of Protein Molecules” open=”no”]
[title size=”3″]Abstract[/title]
We present force-clamp data on the collapse of ubiquitin polyproteins from a highly extended state to the folded length, in response to a quench in the force from 110 pN to 5 or 10 pN. Using a recent method for free-energy reconstruction from the observed nonequilibrium trajectories, we find that their statistics is captured by simple diffusion along the end-to-end length. The estimated diffusion coefficient of ∼100 nm² s⁻¹ is significantly slower than expected from viscous effects alone, possibly because of the internal degrees of freedom of the protein. The free-energy profiles give validity to a physical model in which the multiple protein domains collapse all at once and the role of the force is approximately captured by the Bell model.
[title size=”4″]Citation[/title]
Lannon, H.; Haghpanah, J. S.; Montclare, J. K.; Vanden-Eijnden, E.; Brujic, J. Force-Clamp Experiments Reveal the Free-Energy Profile and Diffusion Coefficient of the Collapse of Protein Molecules. Phys. Rev. Lett. 2013, 110, 128301. doi: 10.1103/PhysRevLett.110.128301 [/toggle]
[toggle title=”Anisotropic nanocrystal arrays organized on protein lattices formed by recombinant clathrin fragments” open=”no”]
[title size=”3″]Abstract[/title]
Recombinant clathrin protein fragments form assemblies that template gold nanocrystals in an array across the latticed surface. The nanocrystals exhibit unusual anisotropic morphologies with long range ordering, both of which are dependent upon the presence of a hexahistidine tag on the clathrin heavy chain fragments.
[title size=”4″]Citation[/title]
Hom, N.; Mehta, K. R.; Chou, T.; Foraker, A. B.; Brodsky, F. M.; Kirshenbaum, K.; Montclare, J. K. Anisotropic nanocrystal arrays organized on protein lattices formed by recombinant clathrin fragments. J. Mater. Chem., 2012, 22, 23335–23339. doi: 10.1039/C2JM35019J [/toggle]
[toggle title=”Modulating Supramolecular Assemblies and Mechanical Properties of Engineered Protein Materials by Fluorinated Amino Acids” open=”no”]
[title size=”3″]Abstract[/title]
Here we describe the biosynthesis and characterization of fluorinated protein block polymers comprised of the two self-assembling domains (SADs): elastin (E) and the coiled-coil region of cartilage oligomeric matrix proteins (C). Fluorination is achieved by residue-specific incorporation of p-fluorophenylalanine (pFF) to create pFF-EC, pFF-CE, and pFF-ECE. Global fluorination results in downstream effects on the temperature-dependent secondary structure, supramolecular assembly, and bulk mechanical properties. The impact of fluorination on material properties also differs depending on the orientation of the block configurations as well as the number of domains in the fusion. These studies suggest that integration of fluorinated amino acids within protein materials can be employed to tune the material properties, especially mechanical integrity.
[title size=”4″]Citation[/title]
Yuvienco, C.; More, H. T.; Haghpanah, J. S.; Tu, R. S.; Montclare, J. K. Modulating Supramolecular Assemblies and Mechanical Properties of Engineered Protein Materials by Fluorinated Amino Acids. Biomacromolecules 2012, 13 (8), 2273–2278. doi: 10.1021/bm3005116 [/toggle]
[toggle title=”Effects of Divalent Metals on Nanoscopic Fiber Formation and Small Molecule Recognition of Helical Proteins” open=”no”]
[title size=”3″]Abstract[/title]
Metal dependent protein-based assemblies derived from the cartilage oligomeric matrix protein (C) coiled-coil domain (His6-C) and two variants with mutation at position 40 (His6-T40A) and 44 (His6-L44A) are explored. All proteins have an N-terminal hexahistidine tag (His6) that interacts with divalent metal ions Zn(II) and Ni(II). Binding to Zn(II) confers enhanced helical structure and stability, while Ni(II) promotes aggregation. Surprisingly, His6-L44A undergoes a conformational switch from unstructured to α-helix in the presence of Zn(II). Both His6-C and His6-T40A further assemble into discrete nanofibers that appear to be stabilized by Zn(II) in which the fiber formation is dictated by the α-helical content. Because Ni(II) promotes aggregation, the proteins visibly cluster, forming large fiber mats in the case of His6-C and His6-T40A or aggregated structures as observed for His6-L44A. Due to the unique pentameric assembly of the proteins, recognition of a small molecule within the pore is assessed using curcumin as the guest molecule. In the presence of Zn(II), there is enhanced binding to curcumin, while the addition of Ni(II) causes a loss in binding. It is shown that metal binding serves as a trigger to control the conformation of the proteins, affecting the nanoscopic fibrous assemblies and small molecule recognition abilities.
[title size=”4″]Citation[/title]
[cite]10.1002/adfm.201101627[/cite][/toggle]
[toggle title=”Characterization and identification of the protein partners of Fn3 domain in FnTm2″ open=”no”]
[title size=”3″]Abstract[/title]
Recently, a novel transmembrane protein was found to be up-regulated in the auditory learning pathway of birds and mammals. The protein, FnTm2, was predicted to have an extracellular fibronectin III (Fn3) domain and a single transmembrane domain. By contrast to other studied Fn3 domains the extracellular domain of FnTm2 bears several cysteine residues, which are predicted to form disulfide bonds. The Fn3 domain of the FnTm2 protein was expressed in DH5-α Escherichia coli (E. coli) cells, purified and characterized by circular dichroism (CD). In order to identify binding partners to Fn3, the isolated protein was incubated with bird brain lysate for a pull down treatment. Of the proteins recognized, myelin basic protein (MBP) was identified as a bona fide partner; it was further characterized for binding to Fn3 in vitro via fluorescence spectroscopy and confirmed via isothermal calorimetry (ITC).
[title size=”4″]Citation[/title]
Gunasekar, S. K.; Anjia, L.; Matsui, H.; Montclare, J. K. Effects of Divalent Metals on Nanoscopic Fiber Formation and Small Molecule Recognition of Helical Proteins. Adv. Funct. Mater. 2012, 22 (10), 2154–2159. doi: 10.1016/j.pep.2011.08.026 [/toggle]
[toggle title=”Identification and comparison of cutinases for synthetic polyester degradation” open=”no”]
[title size=”3″]Abstract[/title]
Cutinases have been exploited for a broad range of reactions, from hydrolysis of soluble and insoluble esters to polymer synthesis. To further expand the biotechnological applications of cutinases for synthetic polyester degradation, we perform a comparative activity and stability analysis of five cutinases from Alternaria brassicicola (AbC), Aspergillus fumigatus (AfC), Aspergillus oryzae (AoC), Humicola insolens (HiC), and the well-characterized Fusarium solani (FsC). Of the cutinases, HiC demonstrated enhanced poly(ε-caprolactone) hydrolysis at high temperatures and under all pH values, followed by AoC and AfC. Both AbC and FsC are least stable and function poorly at high temperatures as well as at acidic pH conditions. Surface charge calculations and phylogenetic analysis reveal two important modes of cutinase stabilization: (1) an overall neutral surface charge within the “crowning area” by the active site and (2) additional disulfide bond formation. These studies provide insights useful for reengineering such enzymes with improved function and stability for a wide range of biotransformations.
[title size=”4″]Citation[/title]
Baker, P. J.; Poultney, C.; Liu, Z.; Gross, R.; Montclare, J. K. Identification and comparison of cutinases for synthetic polyester degradation. Appl. Microbiol. Biotechnol. 2012, 93, 229–240. doi: 10.1007/s00253-011-3402-4 [/toggle]
[toggle title=”Artificial Protein Block Polymer Libraries Bearing Two SADs: Effects of Elastin Domain Repeats” open=”no”]
[title size=”3″]Abstract[/title]
We have generated protein block polymer EnC and CEn libraries composed of two different self-assembling domains (SADs) derived from elastin (E) and the cartilage oligomeric matrix protein coiled-coil (C). As the E domain is shortened, the polymers exhibit an increase in inverse transition temperature (Tt); however, the range of temperature change differs dramatically between the EnC and CEn library. Whereas all polymers assemble into nanoparticles, the bulk mechanical properties of the EnC are very different from CEn. The EnC members demonstrate viscolelastic behavior under ambient conditions and assemble into elastic soft gels above their Tt values. By contrast, the CEn members are predominantly viscous at all temperatures. All library members demonstrate binding to curcumin. The differential thermoresponsive behaviors of the EnC and CEn libraries in addition to their small molecule recognition abilities make them suitable for potential use in tissue engineering and drug delivery.
[title size=”4″]Citation[/title]
Dai, M.; Haghpanah, J.; Singh, N.; Roth, E. W.; Liang, A.; Tu, R. S.; Montclare, J. K. Artificial Protein Block Polymer Libraries Bearing Two SADs: Effects of Elastin Domain Repeats. Biomacromolecules 2011, 12 (12), 4240–4246. doi: 10.1021/bm201083d [/toggle]
[toggle title=”Modulating substrate specificity of histone acetyltransferase with unnatural amino acids” open=”no”]
[title size=”3″]Abstract[/title]
Controlling the substrate specificity of enzymes is a major challenge for protein engineers. Here we explore the effects of residue-specific incorporation of ortho-, meta- and para-fluorophenylalanine (oFF, mFF, pFF) on the selectivity of human histone acetyltransferase (HAT) protein, p300/CBP associated factor (PCAF). Varying the position of the fluorine group in the phenylalanine ring confers different effects on the ability of PCAF to acetylate target histone H3 as well as non-histone p53. Surprisingly, pFF–PCAF exhibits an increase in activity for non-histone p53, while mFF–PCAF is selective for histone H3. These results suggest that global incorporation of unnatural amino acids may be used to re-engineer protein specificity.
[title size=”4″]Citation[/title]
Mehta, K. R.; Yang, C. Y.; Montclare, J. K. Modulating substrate specificity of histone acetyltransferase with unnatural amino acids. Mol. BioSyst., 2011, 7, 3050–3055. doi: 10.1039/C1MB05148B [/toggle]
[toggle title=”Development and Implementation of High School Chemistry Modules Using Touch-Screen Technologies” open=”no”]
[title size=”3″]Abstract[/title]
Technology was employed to motivate and captivate students while enriching their in-class education. An outreach program is described that involved college mentors introducing touch-screen technology into a high school chemistry classroom. Three modules were developed, with two of them specifically tailored to encourage comprehension of molecular bonding principles using a chemistry-based iPad app. Feedback-oriented lessons were utilized to pinpoint and address the students’ learning needs and preferences. Integration of the touch-screen technology with the chemistry curriculum demonstrated favorable results for all people involved: the high school teacher received assistance in the classroom, the college mentors gained experience as well as encouraged the high school students to further pursue chemical education, and the high school students received reinforcement in their chemistry curriculum.
[title size=”4″]Citation[/title]
Lewis, M. S.; Zhao, J.; Montclare, J. K. Development and Implementation of High School Chemistry Modules Using Touch-Screen Technologies. J. Chem. Educ. 2012, 89 (8), 1012–1018. doi: 10.1021/ed200484n [/toggle]
[toggle title=”Enhanced Refoldability and Thermoactivity of Fluorinated Phosphotriesterase” open=”no”]
[title size=”3″]Abstract[/title]
Teflon-like globular proteins: Global incorporation of p-fluorophenylalanine (pFF) in the S phosphotriesterase dimer (see figure) was found to stabilize the protein and led to refoldability and improved activity for substrates at elevated temperatures.
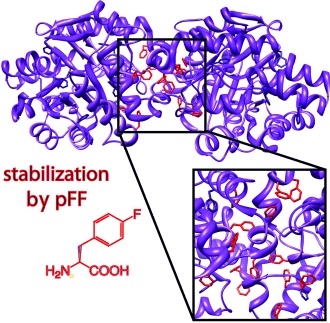
[title size=”4″]Citation[/title]
Baker, P. J. and Montclare, J. K. Enhanced Refoldability and Thermoactivity of Fluorinated Phosphotriesterase. ChemBioChem 2011, 12 (12), 1845–1848. doi: 10.1002/cbic.201100221 [/toggle]
[toggle title=”Implementing and evaluating mentored Chemistry-Biology technology lab modules to promote early Interest in science” open=”no”]
[title size=”3″]Abstract[/title]
Chemistry has become increasingly multidisciplinary. Starting in middle school and high school, however, the different disciplines are taught as distinct subjects and little effort is made to emphasize overlapping concepts. Development of innovative approaches to teaching science through the integration of technology is needed to provide students with a better learning experience that embraces multiple disciplines. Here, we describe an outreach and mentoring program between college and 7th grade students to: (i) encourage 7th grade students to pursue science and engineering degrees; (ii) provide the college students with mentoring experience; and (iii) assist teachers with state-of-the-art educational tools that enhance their teaching.
[title size=”4″]Citation[/title]
Chan, Y. M.; Hom, W.; Montclare, J. K. Implementing and Evaluating Mentored Chemistry−Biology Technology Lab Modules To Promote Early Interest in Science. J. Chem. Educ. 2011, 88 (6), 751–754. doi: 10.1021/ed100476e [/toggle]
[toggle title=”College-Mentored Polymer/Materials Science Modules for Middle and High School Students” open=”no”]
[title size=”3″]Abstract[/title]
Polymers are materials with vast environmental and economic ramifications, yet are generally not discussed in secondary education science curricula. We describe a program in which college mentors develop and implement hands-on, polymer-related experiments to supplement a standard, state regents-prescribed high school chemistry course, as well as a middle school elective course on polymers. Interactive experimentation and feedback-oriented design are highlighted as critical elements to the success of the program. The experiments have been executed in two vastly different institutions: a New York City magnet high school for underprivileged females, and a private middle school for privileged male and female students; the similarities and differences are juxtaposed.
[title size=”4″]Citation[/title]
Lorenzini, R. G.; Lewis, M. S.; Montclare, J. K. College-Mentored Polymer/Materials Science Modules for Middle and High School Students. J. Chem. Educ. 2011, 88 (8), 1105–1108. doi: 10.1021/ed1005618 [/toggle]
[toggle title=”Modified Tat peptide with cationic lipids enhances gene transfection efficiency via temperature-dependent and caveolae-mediated endocytosis” open=”no”]
[title size=”3″]Abstract[/title]
The HIV-1 Tat peptide has been successfully used for intracellular gene delivery. Likewise, various lipid-based methods have shown increased endocytosis and can influence endosomal escape. This study combines the favorable properties of Tat peptide with that of lipid systems for DNA delivery. We combined the lipid FuGENE HD (FH) with the Tat peptide sequence modified with histidine and cysteine residues (mTat). mTat/FH transfection was evaluated by luciferase expression plasmid in five cell types. mTat/FH produced significant improvement in transfection efficiency of all cell lines when compared to FH or mTat. Treatment with chloroquine, associated with energy-dependent endocytosis, significantly increased transfection efficiency with mTat/FH while incubation at low temperature decreased it. The zeta potential of mTat/FH/DNA was significantly higher compared to FH, mTat, or their DNA combination in the presence of serum, and it was correlated with transfection efficiency. The particle size of the FH/DNA complex was significantly reduced by addition of mTat. Filipin III, an inhibitor of caveolae-mediated endocytosis, significantly inhibited mTat/FH transfection, but transfection was increased by chlorpromazine, an inhibitor of clathrin-mediated endocytosis. These findings demonstrated the feasibility of using a combination of mTat with lipids, utilizing temperature-dependent and caveolae-mediated endocytosis, as a potentially attractive non-viral gene vector.
[title size=”4″]Citation[/title]
Yamano, S.; Dai, J.; Yuvienco, C.; Khapli, S.; Moursi, A. M.; Montclare, J. K. Modified Tat peptide with cationic lipids enhances gene transfection efficiency via temperature-dependent and caveolae-mediated endocytosis. J. Control. Release 2011, 152 (2), 278–285. doi: 10.1016/j.jconrel.2011.02.004 [/toggle]
[toggle title=”Mutagenesis of tGCN5 core region reveals two critical surface residues F90 and R140″ open=”no”]
[title size=”3″]Abstract[/title]
Tetrahymena General Control Non-Derepressor 5 (tGCN5) is a critical regulator of gene transcription via acetylation of histones. Since the acetylation ability has been attributed to the “core region”, we perform mutagenesis of residues within the tGCN5 “core region” in order to identify those critical for function and stability. Residues that do not participate in catalysis are identified, mutated and characterized for activity, structure and thermodynamic stability. Variants I107V, Q114L, A121T and A130S maintain the acetylation function relative to wild-type tGCN5, while variants F90Y, F112R and R140H completely abolish function. Of the three non-functional variants, since F112 is mutated into a non-homologous charged residue, a loss in function is expected. However, the remaining two variants are mutated into homologous residues, suggesting that F90 and R140 are critical for the activity of tGCN5. While mutation to homologous residue maintains acetylation of histone H3 for the majority of the variants, the two surface-exposed residues, F90 and R140, appear to be essential for tGCN5 function, structure or stability.
[title size=”4″]Citation[/title]
Mehta, K. R.; Chan, Y. M.; Lee, M. X.; Yang, C. Y.; Voloshchuka, N.; Montclare, J. K. Mutagenesis of tGCN5 core region reveals two critical surface residues F90 and R140. Biochem. Biophys. Res. Commun. 2010, 400 (3), 363–368. doi: 10.1016/j.bbrc.2010.08.069 [/toggle]
[toggle title=”Supramolecular assembly and small molecule recognition by genetically engineered protein block polymers composed of two SADs” open=”no”]
[title size=”3″]Abstract[/title]
Genetically engineered protein block polymers are an important class of biomaterials that have gained significant attention in recent years due to their potential applications in biotechnology, electronics and medicine. The majority of the protein materials have been composed of at least a single self-assembling domain (SAD), enabling the formation of supramolecular structures. Recently, we developed block polymers consisting of two distinct SADs derived from an elastin-mimetic polypeptide (E) and the alpha-helical COMPcc (C). These protein polymers, synthesized as the block sequences—EC, CE, and ECE—were assessed for overall conformation and macroscopic thermoresponsive behavior. Here, we investigate the supramolecular assembly as well as the small molecule binding and release profile of these block polymers. Our results demonstrate that the protein polymers assemble into particles as well as fully or partially networked structures in a concentration dependent manner that is distinct from the individual E and C homopolymers and the E+C non-covalent mixture. In contrast to synthetic block polymers, the structured assembly, binding and release abilities are highly dependent on the composition and orientation of the blocks. These results reveal the promise for these block polymers for therapeutic delivery and biomedical scaffolds.
[title size=”4″]Citation[/title]
Haghpanah, J. S.; Yuvienco, C.; Roth, E. W.; Liang, A.; Tu, R. S.; Montclare, J. K. Supramolecular assembly and small molecule recognition by genetically engineered protein block polymers composed of two SADs. Mol. BioSyst. 2010, 6, 1662–1667. doi: 10.1039/C002353A [/toggle]
[toggle title=”Biotransformations Using Cutinase” open=”no”]
[title size=”3″]Abstract[/title]
There is a growing interest in sustainable and environmentally-friendly solutions for the industrial manufacturing of chemicals to replace non-renewable fossil fuel-based feedstocks. Biotransformations provide an alternative methodology to traditional reactions by taking advantage of the biochemical diversity of microorganisms to provide a chemo-, regio- and enantioselectivity, which are not always available via traditional synthetic approaches. Cutinases are enzymes secreted from phytopathogens and have been proven to be useful for several different biotransformations. This review will investigate the structure and function of cutinase. Further, it will detail cutinase activity towards both natural and non-natural substrates as well as methods employed to impart stability. Finally, we will describe the different role that cutinase has played in biotransformation reactions for biotechnology applications.
[title size=”4″]Citation[/title]
Baker, P. J. and Montclare, J. K. Biotransformations Using Cutinase. In Green Polymer Chemistry: Biocatalysis and Biomaterials ACS symposium series, vol. 1043; American Chemical Society: Washington, DC, 2010; pp. 141-158. doi: 10.1021/bk-2010-1043.ch011 [/toggle]
[toggle title=”Incorporation of Unnatural Amino Acids for Synthetic Biology” open=”no”]
[title size=”3″]Abstract[/title]
The challenge of synthetic biology lies in the construction of artificial cellular systems. This requires the development of modular “parts” that can be integrated into living systems to elicit an artificial, yet programmed, response or function. The development of methods to engineer proteins bearing unnatural amino acids (UAAs) provides essential components that may address this challenge. Here we review the emerging strategies for incorporating UAAs into proteins with the endgame of engineering artificial cells and organisms.
[title size=”4″]Citation[/title]
Voloshchuka, N. and Montclare, J. K. Incorporation of unnatural amino acids for synthetic biology. Mol. BioSyst. 2010, 6, 65–80. doi: 10.1039/B909200P [/toggle]
[/accordian]
[/tab]
[tab id=3]
[accordian]
[toggle title=”Structural and Functional Studies of Aspergillus oryzae Cutinase: Enhanced Thermostability and Hydrolytic Activity of Synthetic Ester and Polyester Degradation” open=”yes”]
[title size=”3″]Abstract[/title]
Cutinases are responsible for hydrolysis of the protective cutin lipid polyester matrix in plants and thus have been exploited for hydrolysis of small molecule esters and polyesters. Here we explore the reactivity, stability, and structure of Aspergillus oryzae cutinase and compare it to the well-studied enzyme from Fusarium solani. Two critical differences are highlighted in the crystallographic analysis of the A. oryzae structure: (i) an additional disulfide bond and (ii) a topologically favored catalytic triad with a continuous and deep groove. These structural features of A. oryzae cutinase are proposed to result in an improved hydrolytic activity and altered substrate specificity profile, enhanced thermostability, and remarkable reactivity toward the degradation of the synthetic polyester polycaprolactone. The results presented here provide insight into engineering new cutinase-inspired biocatalysts with tailor-made properties.
[title size=”4″]Citation[/title]
Liu, Z.; Gosser, Y.; Baker, P. J.; Ravee, Y.; Lu, Z.; Alemu, G.; Li, H.; Butterfoss, G. L.; Kong, X. -P.; Gross, R.; Montclare, J. K. Structural and Functional Studies of Aspergillus oryzae Cutinase: Enhanced Thermostability and Hydrolytic Activity of Synthetic Ester and Polyester Degradation. J. Am. Chem. Soc. 2009, 131 (43), 15711–15716. doi: 10.1021/ja9046697 [/toggle]
[toggle title=”Artificial Protein Block Copolymers Blocks Comprising Two Distinct Self-Assembling Domains” open=”no”]
[title size=”3″]Abstract[/title]
It’s the way that you do it: Block polymers composed of elastin (E) and COMPcc (C) domains (see figure) have been synthesised and characterised. The protein motifs were chosen for their structures and distinct self-assembly modes. We demonstrate that the micro- and macrostructures of the polymers are dictated by the orientation of the fusions and the number of repeated blocks.
[title size=”4″]Citation[/title]
Haghpanah, J. S.; Yuvienco, C.; Civay, D. E.; Barra, H.; Baker, P. J.; Khapli, S.; Voloshchuk, N.; Gunasekar, S. K.; Muthukumar, M.; Montclare, J. K. Artificial Protein Block Copolymers Blocks Comprising Two Distinct Self-Assembling Domains. ChemBioChem 2009, 10 (17), 2733–2735. doi: 10.1002/cbic.200900539 [/toggle]
[toggle title=”N-terminal Aliphatic Residues Dictate the Structure, Stability, Assembly and Small Molecule Binding of the coiled-coil region of COMP” open=”no”]
[title size=”3″]Abstract[/title]
The coiled-coil domain of cartilage oligomeric matrix protein (COMPcc) assembles into a homopentamer that naturally recognizes the small molecule 1,25-dihydroxyvitamin D3 (vit D). To identify the residues critical for the structure, stability, oligomerization, and binding to vit D as well as two other small molecules, all-trans-retinol (ATR) and curcumin (CCM), here we perform an alanine scanning mutagenesis study. Ten residues lining the hydrophobic pocket of COMPcc were mutated into alanine; of the mutated residues, the N-terminal aliphatic residues L37, L44, V47, and L51 are responsible for maintaining the structure and function. Furthermore, two polar residues, T40 and Q54, within the N-terminal region when converted into alanine improve the α-helical structure, stability, and self-assembly behavior. Helical stability, oligomerization, and binding appear to be linked in a manner in which mutations that abolish helical structure and assembly bind poorly to vit D, ATR, and CCM. These results provide not only insight into COMPcc and its functional role but also useful guidelines for the design of stable, pentameric coiled-coils capable of selectively storing and delivering various small molecules.
[title size=”4″]Citation[/title]
Gunasekar, S. K.; Asnani, M.; Limbad, C.; Haghpanah, J. S.; Hom, W.; Barra, H.; Nanda, S.; Lu, M.; Montclare, J. K. N-Terminal Aliphatic Residues Dictate the Structure, Stability, Assembly, and Small Molecule Binding of the Coiled-Coil Region of Cartilage Oligomeric Matrix Protein. Biochemistry 2009, 48 (36), 8559–8567. doi: 10.1021/bi900534r [/toggle]
[toggle title=”Positional effects of monofluorinated phenylalanines on histone acetyltransferase stability and activity” open=”no”]
[title size=”3″]Abstract[/title]
To explore the impact of global incorporation of fluorinated aromatic amino acids on protein function, we investigated the effects of three monofluorinated phenylalanine analogs para-fluorophenylalanine (pFF), meta-fluorophenylalanine (mFF), and ortho-fluorophenylalanine (oFF) on the stability and enzymatic activity of the histone acetyltransferase (HAT), tGCN5. We selected this set of fluorinated amino acids because they bear the same size and overall polarity but alter in side chain shape and dipole direction. Our experiments showed that among three fluorinated amino acids, the global incorporation of pFF affords the smallest perturbation to the structure and function of tGCN5.
[title size=”4″]Citation[/title]
Voloshchuk, N.; Zhu, A. Y.; Snydacker, D.; Montclare, J. K. Positional effects of monofluorinated phenylalanines on histone acetyltransferase stability and activity. Bioorg. Med. Chem. 2009, 19 (18), 5449–5451. doi: 10.1016/j.bmcl.2009.07.093 [/toggle]
[toggle title=”Frozen Cyclohexane-in-Water Emulsion as a Sacrificial Template for the Synthesis of Multilayered Polyelectrolyte Microcapsules” open=”no”]
[title size=”3″]Abstract[/title]
This paper reports the application of frozen cyclohexane-in-water emulsions as sacrificial templates for the fabrication of hollow microcapsules through layer-by-layer assembly of polyelectrolytes, poly(styrenesulfonate sodium salt), and poly(allylamine hydrochloride). Extraction of the cyclohexane phase from frozen emulsions stabilized with 11 polyelectrolyte layers by compatibilization with 30% v/v ethanol leads to the formation of water-filled microcapsules while preserving the spherical geometry. The majority of microcapsules (>90%) are prepared with intact polyelectrolyte membranes as measured by their deformation induced by osmotic pressure. This work provides a new route for the synthesis of hollow multilayered microcapsules under mild operating conditions.
[title size=”4″]Citation[/title]
Khapli, S.; Kim, J. R.; Montclare, J. K.; Levicky, R.; Porfiri, M.; Sofou, S. Frozen Cyclohexane-in-Water Emulsion as a Sacrificial Template for the Synthesis of Multilayered Polyelectrolyte Microcapsules. Langmuir 2009, 25 (17), 9728–9733. doi: 10.1021/la901020j [/toggle]
[toggle title=”Biosynthesis and Stability of Coiled-Coil Peptides Containing (2S,4R)-5,5,5-Trifluoroleucine and (2S,4S)-5,5,5-Trifluoroleucine” open=”no”]
[title size=”3″]Abstract[/title]
Life in stereo: We report the effects of 5,5,5-trifluoroleucine (TFL) stereochemistry on coiled-coil peptide biosynthesis and stability. We demonstrate that two diastereoisomers of TFL are activated and incorporated into peptides expressed in E. coli. Coiled-coil homodimers of these peptides exhibited increased stability. An equimolar mixture of the two fluorinated peptides formed a heterodimer of modestly enhanced thermal stability relative to the homodimers.
[title size=”4″]Citation[/title]
Montclare, J. K.; Son, S.; Clark, G. A.; Kumar, K.; Tirrell, D. A. Biosynthesis and Stability of Coiled-Coil Peptides Containing (2S,4R)-5,5,5-Trifluoroleucine and (2S,4S)-5,5,5-Trifluoroleucine. ChemBioChem 2009, 10 (1), 84–86. doi: 10.1002/cbic.200800164 [/toggle]
[toggle title=”Elastin-Based Protein Polymers” open=”no”]
[title size=”3″]Abstract[/title]
The synthesis of well-defined macromolecular structures with controlled properties is critical for the production of advanced materials for biological and industrial applications. Inspired by nature’s ability to create proteins with exquisite control, we focus on the applications of elastin and elastin-derived polymers for materials design. The elucidation of elastin biochemical, conformational and physical properties offers insight into the fabrication of novel biomaterials. As part of this review, we highlight some of the recent advances that permit the generation of customized elastin-based polymers. These developments provide an added level of control vital to the future construction of tailor-made supramolecular structures with emergent physical, mechanical and biological properties.
[title size=”4″]Citation[/title]
Baker, P. J.; Haghpanah, J. S.; Montclare, J. K. Elastin-Based Protein Polymers. In Polymer Biocatalysis and Biomaterials II ACS Symposium Series Vol. 999; American Chemical Society: Washington D.C., USA, 2008; pp 37–51. doi: 10.1021/bk-2008-0999.ch003 [/toggle]
[toggle title=”Assembly of bioinspired helical protein fibers” open=”no”]
[title size=”3″]Abstract[/title]
Advances in protein and peptide technologies not only enable the study of basic folding and function of natural structures but also the design of novel scaffolds with the ability to form assemblies of varied shapes and sizes. Tremendous progress has been made in our understanding of α-helices in nature especially in the context of the coiled-coil. The information gleaned from investigating coiled- coils has been used to design novel α-helical fibers with prescribed morphology and dimensions. This review focuses on the lessons learned from the assembly of natural coiled-coils and how this knowledge can be used to tailor helical fibers as novel bioinspired materials.
[title size=”4″]Citation[/title]
Gunasekar, S. K.; Haghpanah, J. S.; Montclare, J. K. Assembly of bioinspired helical protein fibers. Polym. Adv. Technol. 2008, 19 (6), 454–468. doi: 10.1002/pat.1136 [/toggle]
[toggle title=”Fluorinated chloramphenicol acetyltransferase thermostability and activity profile: Improved thermostability by a single-isoleucine mutant” open=”no”]
[title size=”3″]Abstract[/title]
A lysate-based thermostability and activity profile is described for chloramphenicol acetyltransferase (CAT) expressed in trifluoroleucine, T (CAT T). CAT and 13 single-isoleucine CAT mutants were expressed in medium supplemented with T and assayed for thermostability on cell lysates. Although fluorinated mutants, L82I T and L208I T, showed losses in thermostability, the L158I T fluorinated mutant demonstrated an enhanced thermostability relative to CAT T. Further characterization of L158I T suggested that T at position 158 contributed to a portion of the observed loss in thermostability upon global fluorination.
[title size=”4″]Citation[/title]
Voloshchuka, N.; Lee, M. X.; Zhu, W. W.; Tanrikulu, I. C.; Montclare, J. K. Fluorinated chloramphenicol acetyltransferase thermostability and activity profile: Improved thermostability by a single-isoleucine mutant. Bioorg. Med. Chem. 2007, 17 (21), 5907–5911. doi: 10.1016/j.bmcl.2007.07.107 [/toggle]
[toggle title=”Influence of global fluorination on chloramphenicol acetyltransferase activity and stability” open=”no”]
[title size=”3″]Abstract[/title]
Varied levels of fluorinated amino acid have been introduced biosynthetically to test the functional limits of global substitution on enzymatic activity and stability. Replacement of all the leucine (LEU) residues in the enzyme chloramphenicol acetyltransferase (CAT) with the analog, 5′,5′,5′-trifluoroleucine (TFL), results in the maintenance of enzymatic activity under ambient temperatures as well as an enhancement in secondary structure but loss in stability against heat and denaturants or organic co-solvents. Although catalytic activity of the fully substituted CAT is preserved under standard reaction conditions compared to the wild-type enzyme both in vitro and in vivo, as the incorporation levels increase, a concomitant reduction in thermostability and chemostability is observed. Circular dichroism (CD) studies reveal that although fluorination greatly improves the secondary structure of CAT, a large structural destabilization upon increased levels of TFL incorporation occurs at elevated temperatures. These data suggest that enhanced secondary structure afforded by TFL incorporation does not necessarily lead to an improvement in stability.
[title size=”4″]Citation[/title]
Panchenko, T.; Zhu, W. W.; Montclare, J. K. Influence of global fluorination on chloramphenicol acetyltransferase activity and stability. Biotechnol. Bioeng. 2006, 94 (5), 921–930. doi: 10.1002/bit.20910 [/toggle]
[toggle title=”Evolving Proteins of Novel Composition” open=”no”]
[title size=”3″]Abstract[/title]
Changing its nature: Global incorporation of noncanonical amino acids can alter the behavior of proteins in useful ways. In some cases, however, replacement of natural amino acids by noncanonical analogues (blue bars in picture) can cause loss of protein stability. After several generations, functional proteins of non-natural composition were prepared through residue-specific incorporation combined with directed evolution.
[title size=”4″]Citation[/title]
Montclare, J. K. and Tirrell, D. A. Evolving Proteins of Novel Composition. Angew. Chem. Int. Ed. 2006, 45 (27), 4518–4521. doi: 10.1002/ange.200600088 [/toggle]
[/accordian]
[/tab]
[/tabs]
[/three_fourth]